Health Systems Face Growing Gap in Cell and Gene Therapy Implementation
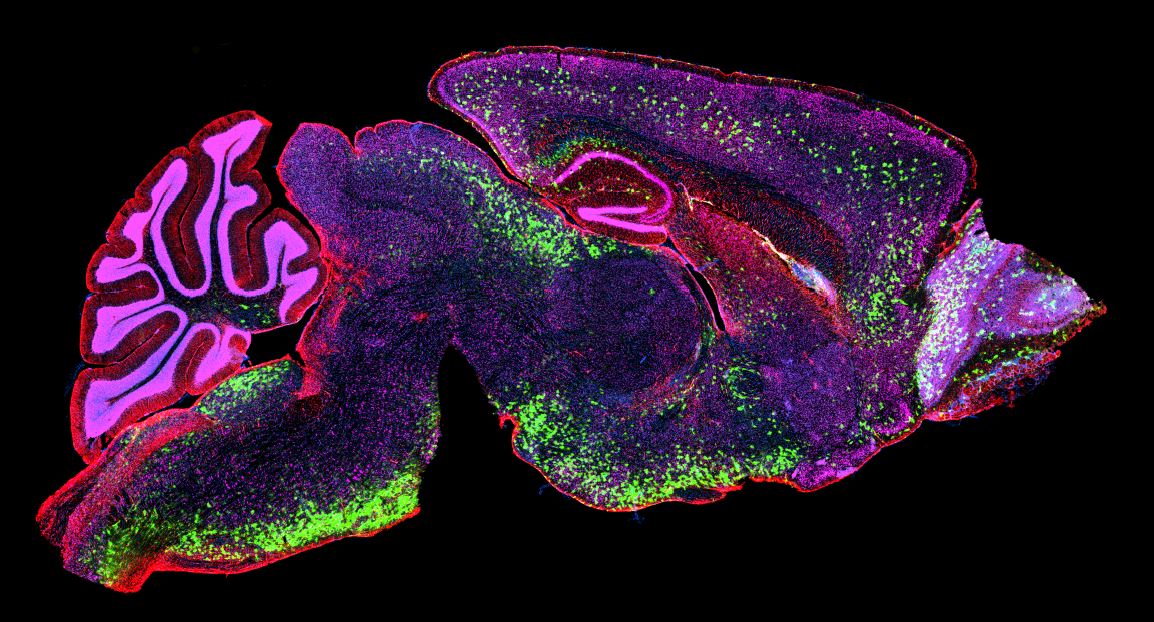
Health Systems Face Growing Gap in Cell and Gene Therapy Implementation https://pediatricsnationwide.org/wp-content/uploads/2026/04/AdobeStock_221145581-1024x768.jpeg 1024 768 Pam Georgiana https://pediatricsnationwide.org/wp-content/uploads/2023/07/May-2023.jpgNew manuscript outlines practical strategies to address safety, operational and financial challenges. As cell and gene therapies (CGTs) are increasingly incorporated into standard clinical care, health systems face a widening operational gap. An invited manuscript published in Transfusion outlines the challenge and presents a practical framework, informed by the experience at Nationwide Children’s, to guide…