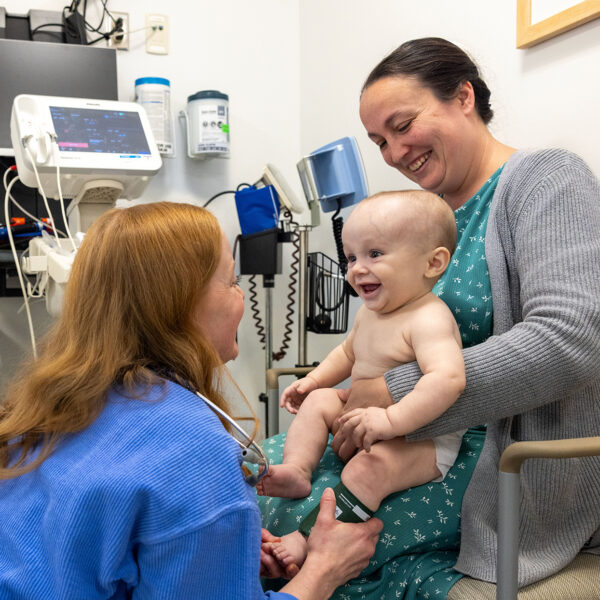Nationwide Children’s Hospital Completes One of the World’s First Prenatal Treatments for Spinal Muscular Atrophy
Nationwide Children’s Hospital Completes One of the World’s First Prenatal Treatments for Spinal Muscular Atrophy https://pediatricsnationwide.org/wp-content/uploads/2026/02/Original_hi_res_JPG-110425SC0431_Edit-1024x600.jpg 1024 600 Katie Brind'Amour, PhD, MS, CHES Katie Brind'Amour, PhD, MS, CHES https://pediatricsnationwide.org/wp-content/uploads/2021/03/Katie-B-portrait.gif
Investigational third-trimester, transplacental “bridging” therapy with risdiplam — followed by rapid postnatal treatment — aims to protect motor neurons during a critical window before and immediately after birth.
When Stacy Auker found out she was pregnant with her third child, she knew a diagnosis of spinal muscular atrophy (SMA) was a possibility.
“We have relatives who have been affected, so I got tested during pregnancy with each of my kids,” Auker explains.
Her baby on the way tested positive for SMA type 1 — a severe neurodegenerative genetic condition that typically results in death in early childhood. Despite the news, Auker and her husband, Clyde, remained optimistic.
“There are good treatments now that we knew could help our baby,” says Auker, referring to the small molecule therapy risdiplam, an oral medication that increases production of full-length SMN protein by modifying SMN2 splicing, and a gene therapy called ZOLGENSMA®, a one-time gene therapy that delivers a functional copy of the SMN1 gene to support SMN protein expression.
Auker’s physician referred her to the maternal-fetal medicine specialists at Nationwide Children’s Hospital, where SMA experts led the development of Zolgensma through its approval in 2019. When they met with Auker, the experts in the Fetal Center told her to expect prenatal monitoring and early postnatal initiation of risdiplam and Zolgensma, with follow-up in the SMA Clinic after delivery.
Then, new information shifted the conversation.
“Two weeks later, Dr. Etchegaray read the first case report of prenatal risdiplam for SMA and immediately thought of the Aukers,” says Elizabeth Swartz, RN, BSN, C-EFM, the Fetal Center nurse coordinator who managed their case. “We’re always excited to pursue innovative treatments for our families.”
“I took it to the neurologists in the SMA Clinic and said, ‘Is this crazy? Can we do this?’” says Adolfo Etchegaray, MD, maternal-fetal medicine specialist and chief of Fetal Medicine at Nationwide Children’s.
Once the medical team felt confident they could feasibly provide prenatal treatment and the fetal ethics committee agreed the approach would offer an appropriate balance of maternal and fetal safety, they presented the idea to Auker.
“Dr. Etchegaray called me and asked what I thought about starting treatment before the baby was born,” says Auker, who admits to being nervous at first, but eager to do anything she could to give her child a developmental advantage.
The approach was inspired by a prenatal risdiplam case reported by clinicians at St. Jude Children’s Research Hospital and published in The New England Journal of Medicine, which described encouraging early outcomes through 30 months of age. A second case was subsequently reported by clinicians in Germany, placing the Aukers among a small number of known cases worldwide.

Adolfo Etchegaray, MD
“Cases like these are changing the paradigm of managing some monogenic conditions,” says Dr. Etchegaray. “We used to counsel families primarily about what to expect after birth. Now we recognize that, in many conditions, injury begins during pregnancy. If we have a therapy that helps after birth, we should ask whether starting earlier could help prevent irreversible damage. Sometimes that earlier start can be a practical ‘bridge’ in the third trimester — leveraging transplacental delivery to cover the window before the newborn can reliably receive definitive therapy.”
Upon Auker’s consent, a host of team members including pharmacists, social workers, neonatologists, fetal medicine experts, radiologists, neurologists, geneticists, researchers and nurses got to work. They helped the Aukers obtain insurance and pursued investigational approval from the Food and Drug Administration for use of risdiplam before birth — taken orally by Auker.
The Genentech Patient Foundation provided the drug free of charge, and Auker initiated oral risdiplam at Week 34 of her pregnancy. To ensure the baby could obtain therapy right after birth, Auker began with pills but switched to the liquid formulation (which could be used without delay for the baby after birth) closer to her anticipated delivery date.
Bryson was born spontaneously at 38 weeks’ gestation — apparently healthy and with normal muscle tone, reflexes and motor function.
After birth, starting risdiplam can be delayed by authorization and specialty-pharmacy logistics. Bryson, however, received his first postnatal dose the day after delivery. He then underwent gene therapy at 10 days old, making him one of the youngest patients ever to receive Zolgensma.

Anna Connair, WHNP-BC, CNM
“We have collected many samples to look at medication levels and health indicators in both mom and baby throughout this entire experience,” says Anna Connair, APRN, CNM/WHNP-BC, the nurse-midwife at Nationwide Children’s who collaborated with maternal-fetal specialists, neonatologists and the obstetrics team at The Ohio State University Wexner Medical Center to organize postnatal testing and rapid administration of risdiplam to the newborn. “Hopefully this will give the world information about how much medication passed to baby to help with future dosing. It’s an exciting field—you are always thinking about how you can expand fetal treatments to other conditions and families.”
Dr. Etchegaray and the team presented their findings at the 2025 meeting of the Society for Maternal-Fetal Medicine and plan to publish updated findings in a case series together with the teams from Germany, St. Jude and a handful of other institutions that have since initiated risdiplam therapy prenatally.
“This is an example of how fetal medicine is personalized, preventative and participative,” says Dr. Etchegaray, who acknowledges that there is still much to learn about prenatal intervention for SMA and its long-term outcomes. “We have the parents and many specialists all working together to try to change things even before the baby is born.”
Meanwhile, the SMA Clinic at Nationwide Children’s continues with business as usual for most families.

Megan Waldrop, MD
“More research is needed to determine for whom [prenatal risdiplam] should be standard of care,” says Megan Waldrop, MD, pediatric neurologist and neuromuscular disease specialist in the SMA Clinic at Nationwide Children’s. “We know that approximately half of the predicted Type 1 SMA babies treated within the first few weeks of life do really well. The other half could potentially benefit from prenatal treatment with risdiplam, but questions remain in terms of what gestational age to initiate, if a second agent is needed after birth and, if combination therapy is needed, for how long.”
The Aukers continue to provide samples for research with the Nationwide Children’s Fetal Center maternal-fetal medicine team but now follow up with Dr. Waldrop and other experts in the SMA Clinic. In the meantime, Bryson continues with daily risdiplam and, as of the age of 8 months, shows no signs of SMA.
“His siblings adore him — they love to make him laugh,” says Auker.
The SMA Clinic will continue to check his physical and neurological development indefinitely, along with more than 100 other SMA patients in their care.
“We are hopeful Bryson can run and play with his siblings in the future, Lord willing,” says Auker, who understands this outcome would have been impossible prior to the development of these treatments. “We are so thankful and blessed for these wonderful treatment options and the excellent care and support we received from the Nationwide Children’s team.”
Learn More
Prenatal use of risdiplam remains investigational and requires careful regulatory oversight and ethics review. To learn more about prenatal therapies or talk with an expert, visit the Fetal Center.
About the author
Katherine (Katie) Brind’Amour is a freelance medical and health science writer based in Pennsylvania. She has written about nearly every therapeutic area for patients, doctors and the general public. Dr. Brind’Amour specializes in health literacy and patient education. She completed her BS and MS degrees in Biology at Arizona State University and her PhD in Health Services Management and Policy at The Ohio State University. She is a Certified Health Education Specialist and is interested in health promotion via health programs and the communication of medical information.
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 28, 2014
- Posted In:
- Features
- Patient Story
- Research