Speeding Up Science With Patient-Derived Xenografts
Speeding Up Science With Patient-Derived Xenografts https://pediatricsnationwide.org/wp-content/uploads/2026/04/Nintedanib-Lungs_Intro-Art-header-1024x483.jpg 1024 483 Katie Brind'Amour, PhD, MS, CHES Katie Brind'Amour, PhD, MS, CHES https://pediatricsnationwide.org/wp-content/uploads/2021/03/Katie-B-portrait.gif
A perfect storm of advanced technologies and scientific collaboration opens doors to rapid progress in pediatric oncology research.
The world of pediatric cancer research faces the challenges of small patient numbers, increased ethical considerations, limited funding and poorly classified diseases. In some ways, these challenges reflect the field’s successes over the past half century, which have driven overall 10-year survival rates from just under 59% to nearly 83%, with even higher cure rates in some cancers.
The remaining patients, however, have rare or aggressive diagnoses, and the limited number of scattered cases make traditional clinical trials a logistical ordeal — even if investigators can find the funding.
“Gone are the days where we could take a bunch of models, treat them with a drug and say ‘It cured tumors in mice, let’s bring it to the clinic!’ We don’t have that luxury in pediatric cancer,” explains Filemon Dela Cruz, MD, principal investigator in the Pediatric Research in Oncology Xenografting Consortium (PROXC) and a pediatric hematologist-oncologist at Memorial Sloan Kettering Cancer Center, where PROXC is hosted. “To be able to answer the questions, you need numbers. We’re fortunate that these difficult patients are a small proportion of the patients we treat, but it means we have to be even more selective about what agents we nominate to develop further in the clinic.”
A Meeting of the Minds
PROXC began as a group of clinician-scientists interested in optimizing and standardizing protocols for their own development of patient-derived tumor xenograft models. These models maintain the unique biology and complex, living-tumor heterogeneity of the original patients from whom they were obtained.
At Nationwide Children’s Hospital, these samples are collected through the Bio-KIDS program, a universal patient consent that allows donation of leftover tissue from a clinically indicated surgery or biopsy. Each tumor receives its indicated clinical workup from pathologists, which often includes genomic sequencing. Tissue samples that remain are then placed into specialized mouse models, where they grow into patient-derived tumors and are cataloged according to tumor characteristics, preserving those living tissues for future research.
 “For a long time, the standard lab research model has involved cell lines that were generated 20 years ago and passaged over and over in cell culture. So it’s uncertain how much the cell cultures used today actually look like the tumors that originally came from those patients,” says Ryan Roberts, MD, PhD, physician for the Division of Hematology, Oncology and Blood and Marrow Transplant and principal investigator for the Center for Childhood Cancer Research at the Abigail Wexner Research Institute at Nationwide Children’s. “That is why we have done so much work to try to generate laboratory models that look more like our patients’ cancers, and that represent the heterogeneity of our patients.”
“For a long time, the standard lab research model has involved cell lines that were generated 20 years ago and passaged over and over in cell culture. So it’s uncertain how much the cell cultures used today actually look like the tumors that originally came from those patients,” says Ryan Roberts, MD, PhD, physician for the Division of Hematology, Oncology and Blood and Marrow Transplant and principal investigator for the Center for Childhood Cancer Research at the Abigail Wexner Research Institute at Nationwide Children’s. “That is why we have done so much work to try to generate laboratory models that look more like our patients’ cancers, and that represent the heterogeneity of our patients.”
As a result of efforts such as these, researchers have begun to understand that not all osteosarcomas are alike, nor are all rhabdomyosarcomas or other tumor types. Even within a single patient, tumors are not homogenous. As scientists amass data in different diagnoses, cancers have begun to shift from a single classification, such as medulloblastoma, to subtypes of disease with distinct prognoses and treatments.
“All of these pediatric diseases are going through this process, and having good models helps us figure out which patients could benefit from which drugs,” says Dr. Roberts, who is also one of PROXC’s seven principal investigators, together with researchers at Dana Farber, St. Jude Children’s Research Hospital, Texas Children’s Hospital and University of California San Francisco. “Things are transforming already and will continue to change over the next decade. We are getting much more sophisticated and personalized in prescribing therapy as we learn to use existing drugs more intelligently and develop new drugs for specific biologies.”
After the PROXC constituents developed their protocols for patient-derived xenograft (PDX) creation, they chose to keep their partnership going. They realized that by pooling the unique models they had each developed, they could establish one of the largest PDX repositories in the world.
The group also knew that the consortium could function as a tremendous platform for preclinical research, enabling pediatric oncology studies the likes of which the scientific community had never seen. They all signed a sweeping data- and model-sharing agreement to enable easy exchange of their PDX collections, genomic sequencing data and basic science findings. Then they put their idea to the test.
PDX Progress
Nationwide Children’s alone houses more than 200 models, many of which the entire consortium can now access — some of which are the only models in the world of rare tumor types. Together with contributions from the other member institutions, consortium researchers now have access to more than 700 PDX models.
When someone wishes to study a particular cancer or genotype, they can go to the PROXC “library” to identify all of the group’s relevant models to advance their work. And when a member has a promising therapy to explore, the team has the ability to conduct a multi-site trial with tumor models from numerous patients, using the group’s uniform protocols.
Members even conduct standardized, blinded experiments with drug distribution from a central site. This novel method of preclinical research effectively tests a therapy on tumors from multiple patients, and when done in a blinded fashion, approximates a clinical trial in the most advanced way possible — without involving real patients.
“The gold standard for preclinical work is now patient derived xenografts,” says Dr. Dela Cruz. “The fact that we can do it in a standardized way across multiple sites and models is unprecedented.”
He and a colleague at Memorial Sloane Kettering had seen initial success in cell lines and cell-line-derived xenografts using a repurposed therapy to treat desmoplastic small round cell tumors (DSRCT), an ultra-rare and almost universally fatal type of sarcoma. It wasn’t until they saw the same anti-tumor effect in multiple PDX models of the tumor that they allowed themselves to get excited, though.
“The PDX studies suggested there would be a positive effect, and there was biologic rationale to say this class or type of drug would have activity in this type of tumor,” says Dr. Dela Cruz. “It made sense to move it into a pediatric trial.”
Despite running out of the finite drug supply before the trial could finish accrual, the study had positive results that aligned with preclinical PDX findings — a promising step toward better therapies for DSRCT.
In another PROXC-facilitated success story, Dr. Roberts and his team made inroads into the primary focus of his research: lung-based metastases of osteosarcoma tumors. His findings, reported in Cancer Research, revealed a population of cells that travel to and interact with lung tissue, triggering sustained fibrosis and creating a suitable environment for the growth of new tumors.
Preliminary research in both PDXs and cell-line models suggests that targeting the IL1 signaling from osteosarcoma tumors significantly inhibits metastatic progression. Nintedanib, a drug approved to treat certain types of chronic lung fibrosis and non-small cell lung cancer, showed success in preventing metastasis by inhibiting fibrosis in multiple preclinical investigations.
This preliminary success, sped up considerably by single-cell RNA sequencing and the ability to rapidly and affordably study multiple living tumor models, now faces the same barrier as Dr. Dela Cruz’s: access to the drug for patient trials.
“This is a common issue we run into,” says Dr. Dela Cruz. “We can discover these things, but funding and drug supply are issues that need to be addressed at multiple levels.”
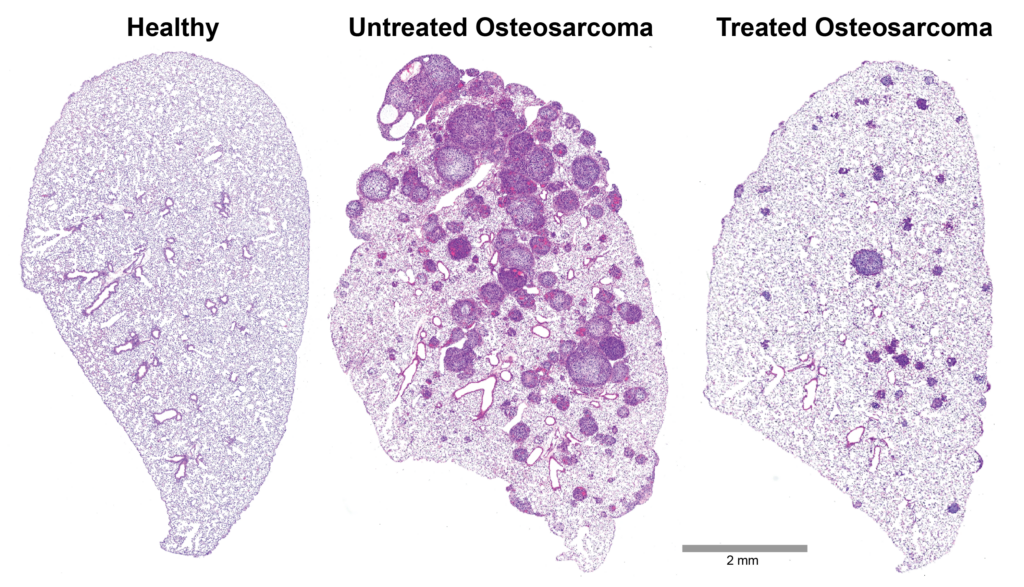
Microscope images of lung from mice with healthy lungs, mice with osteosarcoma bone tumors metastasized to the lungs, and mice with osteosarcoma that were treated with a drug that prevents scar formation in the lung. By blocking the scar-forming reaction, researchers prevented most metastatic lesions from forming. Those that did emerge grew much more slowly. This is the first proof-of-principle step toward a treatment that could save the lives of children and young adults affected by this aggressive disease.
Paving the Way Forward
The PROXC investigators have not let these all-too common barriers deter them from continued progress in pediatric cancer research, and their determination continues to pay dividends.
In a recent Children’s Oncology Group trial, the drug trastuzumab deruxtecan failed in pediatric osteosarcoma. However, because of PDX research (published in Molecular Cancer Therapeutics) suggesting potential efficacy in a different subset of patients, the trial was able to redefine the target population — emphasizing tumors with PDX demonstrated sensitivity regardless of HER2 status, including cases of DSRTC and renal tumors — and redirect the indication to one better informed by tumor biology. Now repositioned, trial enrolment begins again in 2026.
“Without patient-derived xenografts and similar resources, we’re really stuck just studying one patient’s cells. That’s like trying to describe the universe of apples if all you have is a Granny Smith,” says Dr. Roberts. “These models let us appreciate the nuance of what happens in real patients — why some respond and some don’t to a particular drug.
“Without patient-derived xenografts and similar resources, we’re really stuck just studying one patient’s cells. That’s like trying to describe the universe of apples if all you have is a Granny Smith. These models let us appreciate the nuance of what happens in real patients — why some respond and some don’t to a particular drug. Understanding that is hugely important, and we couldn’t do it without these models.”
— Ryan Roberts, MD, PhD
Understanding that is hugely important, and we couldn’t do it without these models.” The world of single-cell sequencing has further opened the world of rare tumors to investigators, shining a light on minority cell populations, mutation similarities across cancers, tumor heterogeneity over time and treatment, and potential weaknesses to exploit with new or existing therapies.
“With single-cell sequencing, I can do an experiment in one week that gives me a body of data that would have taken me 5-6 years to generate less than a decade ago,” says Dr. Roberts. “It’s pretty incredible what these new techniques can do.”
The complexity of these technologies demands expertise in multiple scientific disciplines to bear fruit. At Nationwide Children’s, the Roberts lab relies heavily on the Steve and Cindy Rasmussen Institute for Genomic Medicine for sequencing, the High-Performance Computing Core for data management, the Biopathology Center for overflow storage, the Bio-KIDS program for continued source material, multiple clinical divisions for patient recruitment, the Clinical Trials Office for study logistics and the Animal Tumor Core for help with models. Coupled with a Germain Family Accelerator Grant for sarcoma research and the resources of the full PROXC team, Dr. Roberts is optimistic about what lies ahead for the field of pediatric oncology.
“We can now take all of the cells in a tumor, including the complex networks that form a metastatic lesion, and pick apart all the genes expressed in each individual cell, showing what happens when they are treated with a specific drug,” says Dr. Roberts. “We are trying to use it to design specific solutions that may differ from patient to patient, to pick the best drug for each person and truly deliver precision medicine.”
His team is currently diligently mapping the responses of each enzyme in each cell type for osteosarcoma lung metastases, genome-wide. Once they know which pathways are activated abnormally, they can select or develop drugs that target those pathways.
“We’ve been talking about moving the needle for a long time, but I think we’re at an inflection point because of this unprecedented insight into tumor heterogeneity,” says Dr. Dela Cruz, who believes that computational tools will soon help identify potential sensitivities of rare and resistant tumor types, which can then be reprogrammed or treated to push tumors into a sensitive state.
“This is where PROXC is uniquely equipped to address the remaining challenges in this field,” says Dr. Dela Cruz. “When you have one of the largest PDX collections in the world, you can ask questions no one else can answer.”
This article appeared in the 2026 Spring/Summer print issue. Download the issue here.
References:
- Burke CM, Feinberg TY, Brosius S, Bhanot UK, Jain M, Linkov I, Siddiquee A, Guillan K, Ibañez G, Ndengu AA, Calder P, Rosales N, F Coutinho D, Long MM, Fishkin R, Thyparambil S, Glade Bender JL, Reed DR, You D, Ortiz MV, Slotkin EK, Kung AL, Dela Cruz FS. Anti-tumor activity of trastuzumab deruxtecan in pediatric solid tumors with variable HER2 expression. Molecular Cancer Therapeutics. 2026;25(1):156-167.
- Reinecke JB, Jimenez Garcia L, Gross AC, Cam M, Cannon MV, Gust MJ, Sheridan JP, Gryder BE, Dries R, Roberts RD. Aberrant activation of wound-healing programs within the metastatic niche facilitates lung colonization by osteosarcoma cells. Clinical Cancer Research. 2025 Jan 17;31(2):414-429.
- Reinecke JB, Jimenez Garcia L, Saraf AJ, Hinckley J, Gross AC, Le Pommellet H, Gutpell KM, Cam M, Cannon MV, Gust MJ, Vatelle S, Gryder BE, Dries R, Roberts RD. Metastasis-initiating osteosarcoma subpopulations establish paracrine interactions with lung and tumor cells to create a metastatic niche. Cancer Research. 2025 Nov 14;85(22):4341-4358.
- Slotkin EK, Mauguen A, Dela Cruz FS, Ortiz MV, Avutu V, Meyers PA, Wexler LH, O’Donohue TJ, Kinnaman MD, Kelly CM, D’Angelo SP, Keohan ML, Gounder MM, Thornton K, Nacev BA, Chi P, Rosenbaum E, Dickson M, Pachhal S, Somwar R, Ladanyi M, Robb C, Pandit-Taskar N, Hwang S, Price A, Behr G, Reed DR, Kentsis A, Kung AL, Bender JG, Tap WD. ACR-368, a CHK1/2 kinase inhibitor, in patients with relapsed or refractory desmoplastic small round cell tumor: Phase I/II trial results. JCO Oncology Advances. 2025 Apr 28;2(1):e2400095.
Image credits: Nationwide Children’s (microscopy images courtesy of the Reynolds Lab)
About the author
Katherine (Katie) Brind’Amour is a freelance medical and health science writer based in Pennsylvania. She has written about nearly every therapeutic area for patients, doctors and the general public. Dr. Brind’Amour specializes in health literacy and patient education. She completed her BS and MS degrees in Biology at Arizona State University and her PhD in Health Services Management and Policy at The Ohio State University. She is a Certified Health Education Specialist and is interested in health promotion via health programs and the communication of medical information.
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 28, 2014







