From Bronchopulmonary Dysplasia to Chronic Obstructive Pulmonary Disease: A Developmental Continuum
From Bronchopulmonary Dysplasia to Chronic Obstructive Pulmonary Disease: A Developmental Continuum https://pediatricsnationwide.org/wp-content/uploads/2026/03/W1909850-Rsrch_PedsNW-SpringSummer2026_Pg-13-1024x774.jpg 1024 774 Yan Hu, PhD Yan Hu, PhD https://pediatricsnationwide.org/wp-content/uploads/2026/03/Original_hi_res_JPG-082925BT22-bio-crop.jpg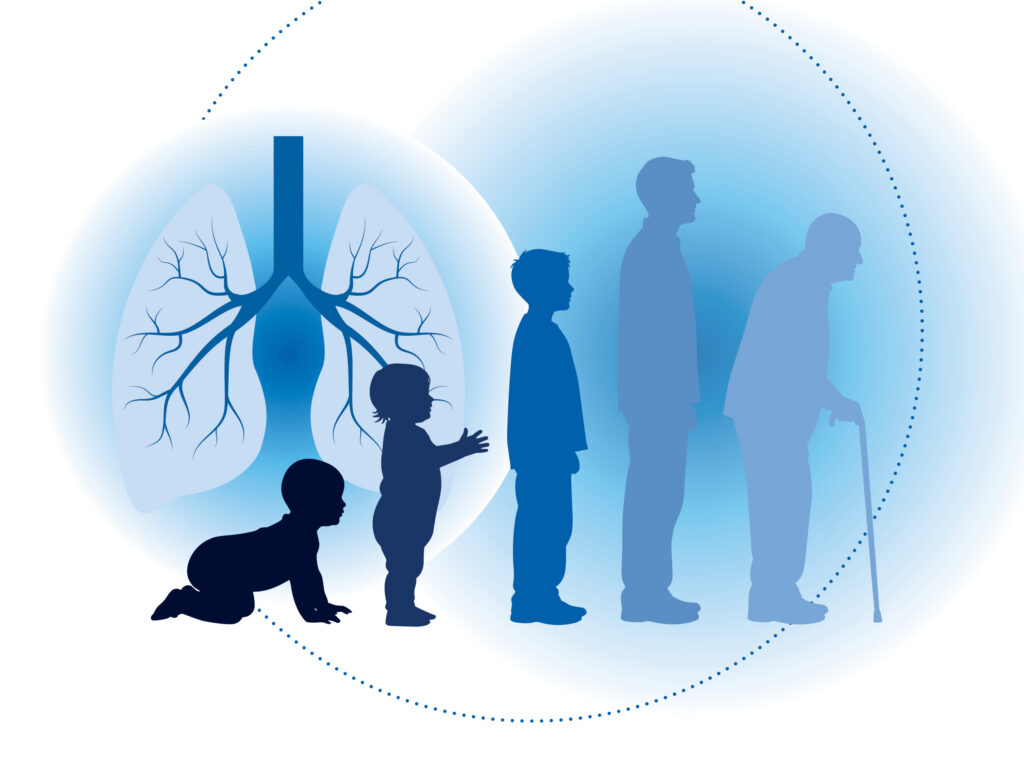
A Prolonged Window of Vulnerability
Lung development is a highly orchestrated process that begins early in gestation and continues well into postnatal life. Following airway branching during the embryonic and pseudoglandular stages, distal lung maturation, including small airway and alveolar formation, extends through infancy and early childhood. This prolonged developmental window renders the lung particularly vulnerable to environmental and inflammatory insults, with lasting consequences for respiratory health.
Premature Birth and the Origins of BPD
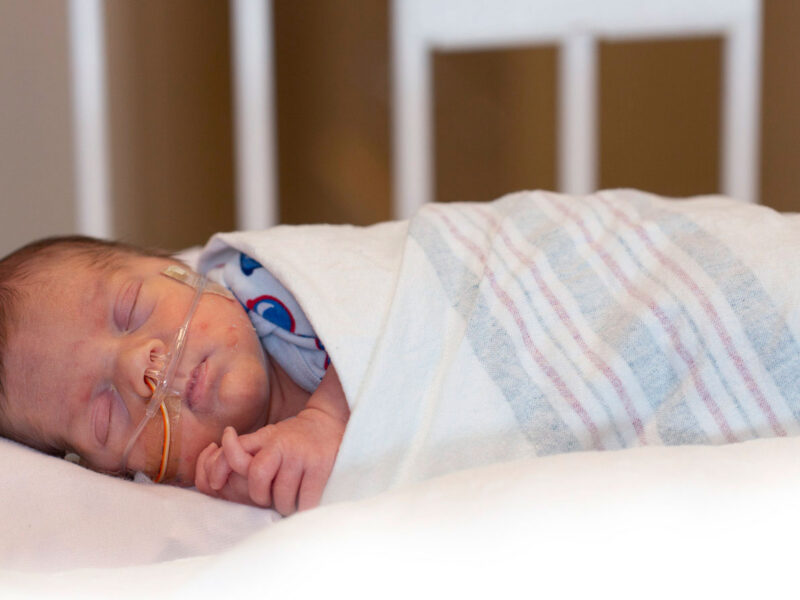
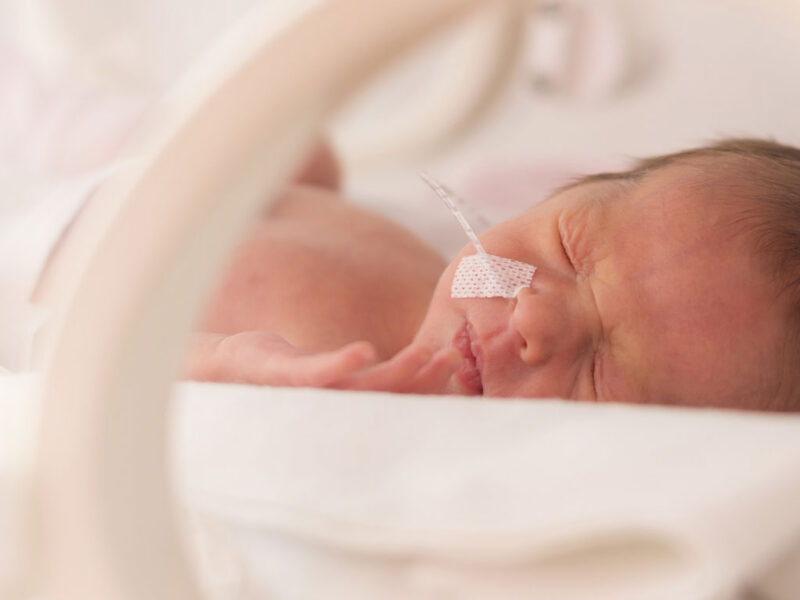
Preterm birth abruptly disrupts this finely tuned program. Exposure of the immature lung to mechanical ventilation, hyperoxia and inflammation occurs during critical windows of epithelial and mesenchymal differentiation, often resulting in bronchopulmonary dysplasia (BPD).
Since its initial description by William H. Northway, Jr., MD, and colleagues in 1967, the clinical phenotype and outcomes of BPD has evolved substantially. Advances in neonatal care, such as antenatal corticosteroids, surfactant replacement and noninvasive ventilation, have dramatically improved survival among extremely preterm infants, including those born before 28 weeks of gestation. However, these successes have shifted the clinical burden from early mortality to long-term respiratory morbidity driven by impaired lung development and regenerative capacity.
A Precursor to Adult Chronic Lung Disease
Increasing evidence now supports the concept that BPD confers increased risk for chronic respiratory morbidity in adulthood, including the development of chronic obstructive pulmonary disease (COPD). Survivors of preterm birth, particularly those with a history of BPD, exhibit persistent airflow limitation, altered architecture and reduced lung function.
These observations challenge the traditional view of COPD as solely an age- or exposure-related disorder and instead position it along a developmental continuum initiated by early-life injury. Shared pathological features, including alveolar simplification, small airway remodeling, chronic inflammation and extracellular matrix dysregulation, suggest common mechanistic roots between BPD and COPD.
Notably, no current therapies effectively promote lung regeneration in either condition, underscoring a critical unmet clinical need.
Regenerative Pathways Across the Lifespan
Building on our work in lung epithelial stem cell dysfunction in COPD, my lab’s research extends beyond a singular focus on alveolar type II cells to investigate a broader spectrum of epithelial progenitor populations, including airway secretory cells capable of contributing to alveolar repair following injury. We leverage high-throughput approaches, including high-resolution spatial transcriptomics and single-cell RNA sequencing, across transgenic animal models and human lung tissues to defi ne molecular programs that govern epithelial cell fate, plasticity and regenerative capacity in health and disease. To functionally interrogate these pathways, we integrate mouse, ferret and human-derived precision-cut lung slices (PCLS), which preserve native tissue architecture and multicellular interactions, alongside lung organoid systems. Together, these complementary platforms enable mechanistic testing of hypotheses generated from transcriptomic analyses and facilitate the identification of targetable pathways that promote effective lung regeneration.
Bridging Developmental Biology and Disease Mechanisms
Despite growing recognition of the developmental origins of chronic lung disease, the biological mechanisms linking early-life injury to adult pathology remain poorly defined. Clinically, the long-term burden of preterm birth and BPD is difficult to quantify due to limited longitudinal follow-up into adulthood. Addressing this gap requires experimental systems that capture both early developmental disruption and long-term regenerative capacity. By integrating models of impaired lung development with subsequent adult injury, and combining these with PCLS and organoid platforms, we aim to better reflect the trajectory from neonatal lung injury to chronic respiratory disease and move toward a precision medicine approach for lifelong lung health.
This article appeared in the 2026 Spring/Summer print issue. Download the issue here.
Reference:
Northway WH Jr, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. New England Journal of Medicine. 1967
Feb 16;276(7):357-368.
Image credit: Nationwide Children’s
About the author
Dr. Hu’s research aims to uncover therapeutic pathways to promote small airway and alveolar regeneration, particularly in diseases where current treatments are limited, such as COPD and BPD. Her lab employs cutting-edge methodologies, including single-cell and spatial transcriptomics, patient-derived lung organoids, ex vivo precision-cut lung slices, and transgenic animal disease models. By integrating these advanced approaches, Dr. Hu seeks to advance our understanding of lung epithelial progenitor cell function and develop innovative strategies to enhance lung tissue repair.
- Posted In:
- Features
- Research
- Second Opinions