Tissue Engineered Vascular Grafts: A Breakthrough for Children With Heart Defects
Tissue Engineered Vascular Grafts: A Breakthrough for Children With Heart Defects https://pediatricsnationwide.org/wp-content/uploads/2026/02/Nationwide_ChildrenΓCOs_TEVG_Final-Flat-1452x1536-1-1-968x1024.jpg 968 1024 Abbie Miller Abbie Miller https://pediatricsnationwide.org/wp-content/uploads/2023/05/051023BT016-Abbie-Crop.jpg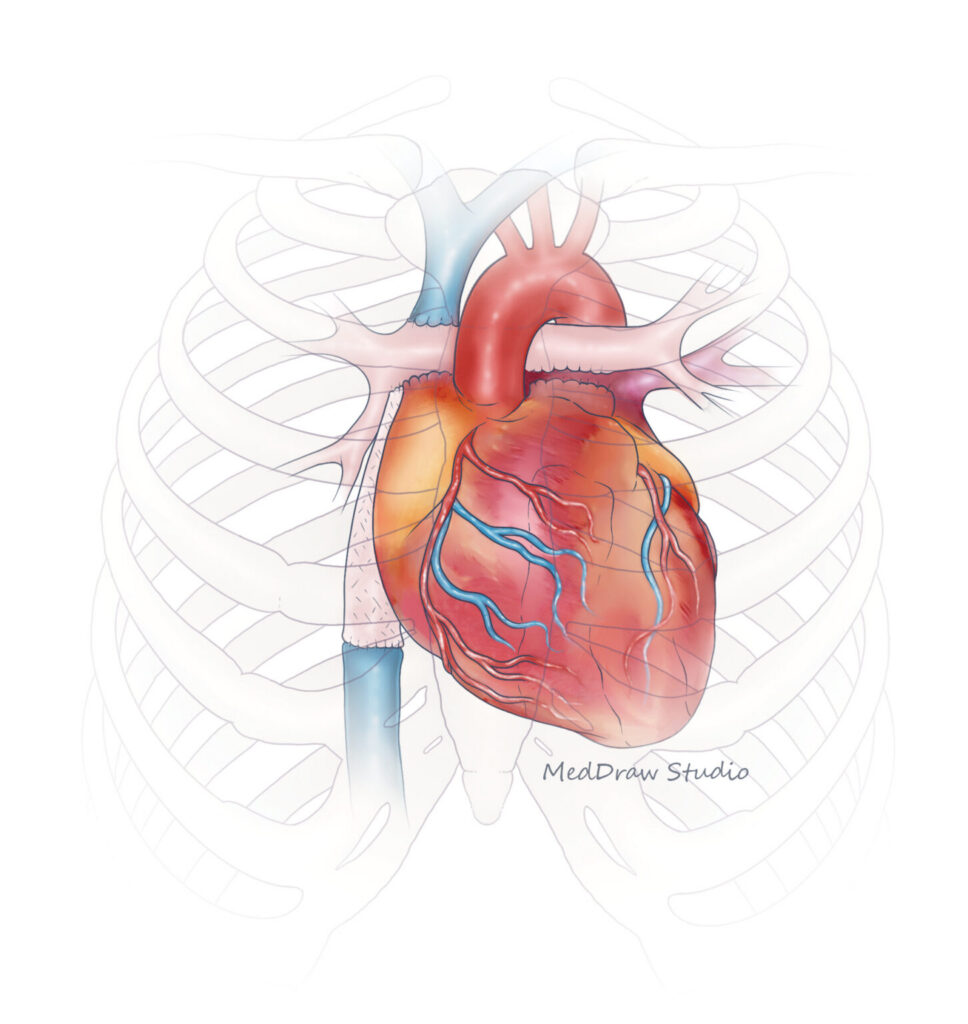
An innovation 30 years in the making is poised to change the way children with single ventricle disease experience life after a Fontan procedure.
Children with single ventricle disease are often described as having “half a heart.” What this really means is that one of their ventricles (either the right or left lower chamber of the heart) is under-developed, making it challenging for the heart to effectively pump blood through the body.
Surgical Advances Require Synthetic Vessels
Advances in surgery enabled many children with single ventricle disease to live into adulthood, with life expectancies growing with advances in care and follow up. When babies are born with single ventricle disease, the standard surgical approach involved three procedures to help the blood flow more efficiently. The third procedure, called the Fontan, is typically performed between the ages of 2 and 5 years old.
In the Fontan, surgeons use a synthetic graft to extend the inferior vena cava (the vessel that brings blood back to the heart from the body) to the pulmonary artery (the vessel that takes blood from the heart to the lungs). Currently, surgeons use a synthetic vessel made from poly(tetrafluoroethylene) (PTFE). However, this vessel does not grow with the child. Considering the average age for the Fontan procedure is 4 years, these children have a lot of growing to do. To keep up with the growing child, additional surgeries to place longer grafts will be needed.
Additionally, these grafts don’t behave like native tissue— that is they don’t stretch or constrict like regular vessels which can lead to problems including clots and stenosis, which is a narrowing of the vessel that restricts blood flow. Treating these complications from synthetic grafts may eventually require stent placement or additional surgeries.
Tissue Engineering a Better Vessel
Christopher Breuer, MD, and Toshiharu Shinoka, MD, PhD, co-directors for the Center for Regenerative Medicine at Nationwide Children’s Hospital, are co-inventors of a tissue engineered vascular graft (TEVG) that could be the answer to the problem of synthetic vessels. Their innovation is nearly 30 years in the making. The Breuer and Shinoka TEVG uses a biodegradable scaffold seeded with the patient’s own cells. After the TEVG is implanted, the scaffold degrades as it is replaced with the child’s own cells, resulting in a neovessel that functions just like a native blood vessel.
“Tissue engineered vascular grafts are superior to other options for pediatric congenital heart patients for several reasons, the most important of which is the graft’s growth capacity,” Dr. Shinoka says. “Our grafts don’t require immunosuppression or anti-rejection medications because they are made up of the patient’s own cells. And they grow with the child, decreasing the number of follow-up surgeries needed with conventional grafts.”
Through a series of preclinical, computational and clinical studies, the team has refined their TEVG and its production.
TEVGs Resist Calcification and Improve Hemodynamics
Calcification is one of the biggest reasons for biomaterial graft failure. In a study published in 2024 in Nature Communications, Dr. Breuer and team showed that compared to expanded PTFE grafts, TEVGs exhibited superior durability, including reduced late-term calcification.
“All of the biomaterials we routinely use for cardiovascular surgery are susceptible to dystrophic calcification,” says Dr. Breuer. “This biomineralization reduces the function of the material, whether it is functioning as a vessel or valve, and can ultimately lead to the need for replacement of the prosthetic.”
Additionally, TEVGs better matched the native vessels in terms of elasticity and responsiveness, which is measured as “compliance.” This better compliance matching allowed the graft to act more like the native vessel in response to changes in blood pressure and flow. This better compliance matching also prevented the formation of stenosis.
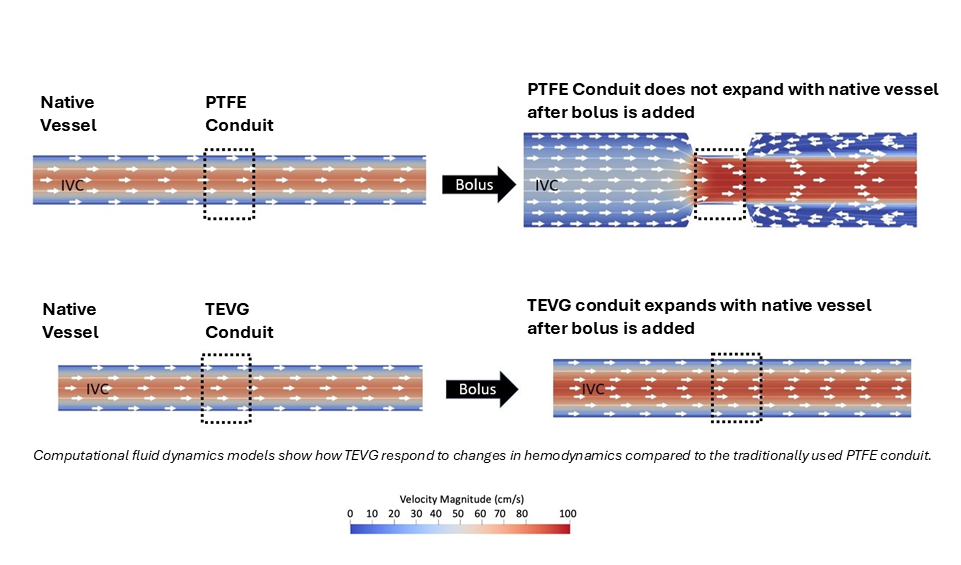
Preventing Stenosis With Surgical Technique
A study published in 2025 in JACC: Basic to Translational Science outlines changes in surgical techniques that would be needed with a future adoption of the tissue engineered vascular graft.
In a large animal study to assess surgical factors that could contribute to stenosis formation, Dr. Breuer and his team implanted and evaluated 50 ovine TEVGs, collecting angiography at 1 and 6 weeks after implantation.
Results indicated that hemodynamics and surgical sizing of the graft were potential driving factors. Regression analysis showed that narrowing at the inflow anastomosis and graft oversizing were significantly correlated with stenosis development. Computational fluid dynamics simulations showed that these factors influenced wall shear stress and flow patterns, contributing to neovessel narrowing. Additionally, clinical trial data supported these findings, emphasizing the importance of matching graft size to the native inflow vessel when using TEVGs.
“This current study demonstrates that, within TEVGs, graft oversizing as well as greater narrowing of the inflow anastomosis at the time of surgery worsen the degree of stenosis,” says Dr. Breuer. “Considering that the current clinical practice is for surgeons to oversize the graft – for good reason, with synthetic grafts you need to give extra room for the child to grow to reduce reoperations – we need to be prepared to incorporate education and behavior change support as we advance the translation and implementation of TEVGs.”
As the TEVGs move closer toward commercialization, Dr. Breuer and his team are thinking about the need to educate surgeons on the best way to use the new materials.
“Our team has been refining our implantation approach for decades, guided by animal studies, clinical trial experience and computational modeling,” says Dr. Breuer. “Sharing what we’ve learned will be an important part of expanding the use of TEVGs if the data continues to support moving forward with the FDA.”
Manufacturing the Grafts: Clean Room vs. Closed System
Just as Drs. Breuer and Shinoka have continued to innovate the graft and the surgical procedures, they have evolved the process to produce the graft.
Currently, the process involves collecting mononuclear cells from the patient’s bone marrow via filtration. Those mononuclear cells are then vacuum seeded onto the biodegradable scaffold.
The idea here is to make the technology safer by eliminating the manual parts of graft production and make it more readily available by eliminating the need for a clean room. Hopefully this would also make the product more accessible for patients.”
– Christopher Breuer, MD, co-director for the Center for Regenerative Medicine at Nationwide Children’s

Once the graft is seeded with cells, the technicians test the graft to ensure all the criteria are met. Once approved, the seeded graft is brought to the operating room for implantation.
“When patients come to the hospital for their operation, they have their bone marrow harvested,” says Dr. Breuer. “Then, we take that bone marrow to a clean room to make the graft. This is a logical and safe way to make the graft, but it presents challenges. Not all hospitals have clean rooms. They’re expensive to build and super expensive to maintain.”
Dr. Breuer and team acknowledge the need for a clean room will ultimately increase the cost of the product and may limit the utility of the technology. So they came up with an alternate option.
“In the next generation version of the graft, one of the things we are working on is making a disposable, closed system, where the whole process could be performed not in a clean room, but in the operating room or in the blood bank where the cells are collected,” explains Dr. Breuer.
The technician injects the bone marrow cells and the process happens in an automated fashion. The cells are filtered and vacuum seeded onto the scaffold in one closed system, eliminating the need for clean room conditions and reducing the number of opportunities for errors in the process.
“The idea here is to make the technology safer by eliminating the manual parts of graft production and make it more readily available by eliminating the need for a clean room,” says Dr. Breuer. “Hopefully this would also make the product more accessible for patients.”
Regardless of whether the graft is made in the clean room or a disposable closed system, the product is the same — a biodegradable scaffold that has been seeded with the patient’s own cells, ready for implantation.

Clean Room

Closed System
Breaking Through to Commercialization
Dr. Breuer and Dr. Shinoka’s TEVG is currently the only one in clinical trials in the United States evaluating their technology in children and has recently been granted Breakthrough designation by the U.S. Food and Drug Administration (FDA). The designation helps speed up the development and review of the device, which could lead to faster access for patients who need it.
The latest clinical trial enrolled their 10th patient in July 2025. “After we follow the 10th patient for 6 months, we’ll review the data. And if it merits, we hope to submit an application to the FDA for clinical approval,” says Dr. Breuer.
In preparation, Dr. Breuer and his team are working with the Office of Technology Commercialization at Nationwide Children’s Hospital. Their goal is to build a startup around the technology to help bring it to market pending FDA approval.
“We’re thrilled to see TEVGs moving from the research labs toward real-world impact through a new startup company, hopefully launching in 2026,” says Kyle Murrah, PhD, senior licensing associate in the Office of Technology Commercialization. “This technology has the potential to transform care for people with complex cardiovascular disease by providing living, growing grafts that can improve outcomes over a lifetime. It’s a powerful example of how innovation at Nationwide Children’s Hospital, combined with the right industry partners, can accelerate life-changing therapies to patients who need them most.”
“I am excited about the prospect of having our first startup in the regenerative medicine space,” adds Margaret Barkett, PhD. “This is an important area of science, and we’re looking forward to bringing this and future regenerative medicine innovations to the marketplace.”
Image Credit: Nationwide Children’s, Courtesy of the Breuer Lab
About the author
Abbie (Roth) Miller, MS, MWC, is a passionate communicator of science. As the manager of medical and science content at Nationwide Children’s Hospital, she shares stories about innovative research and discovery with audiences ranging from parents to preeminent researchers and leaders. She is a Medical Writer Certified®, credentialed by the American Medical Writers Association, and received her masters of science in Health Communication from Boston University.
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Posted In:
- Clinical Updates
- Research