What Happens When Cancer Remission Is in Question?
What Happens When Cancer Remission Is in Question? https://pediatricsnationwide.org/wp-content/uploads/2018/03/leukemia.gif 1024 575 Mary Bates, PhD Mary Bates, PhD https://secure.gravatar.com/avatar/c6233ca2b7754ab7c4c820e14eb518c8?s=96&d=mm&r=g- March 20, 2018
- Mary Bates, PhD
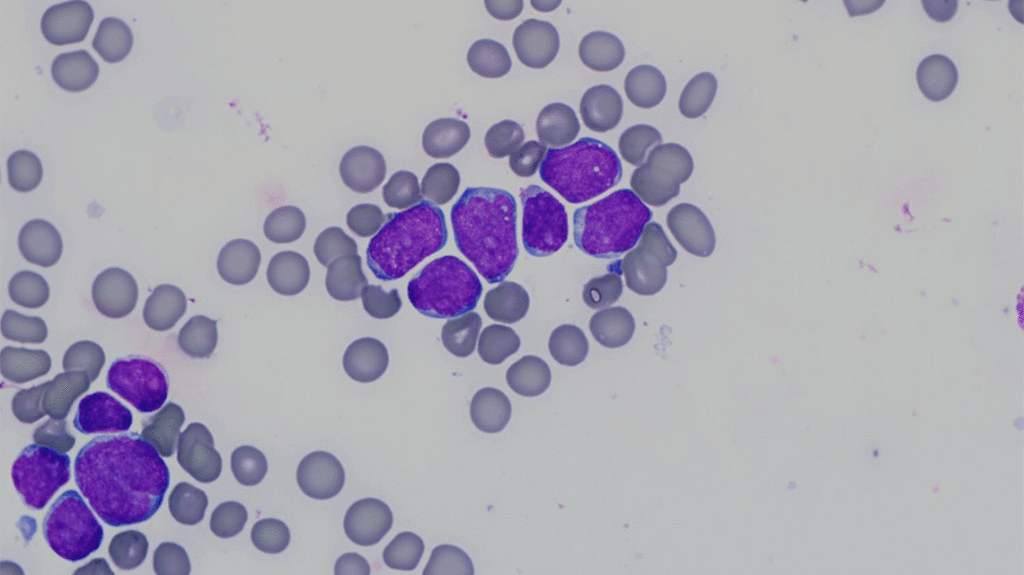
A new study looks at outcomes for children with leukemia when two tests of remission disagree.
In both clinical practice and clinical trials, remission in leukemia is determined by morphological assessment — a person counting cancer cells in a bone marrow sample under a microscope. But more sophisticated technologies, such as flow cytometry or polymerase chain reaction-based techniques, could help overcome many of the limitations of morphological assessments.
Flow cytometry is often used to measure depth of remission after absence of cancer cells has been established microscopically. But what happens when the morphological assessment indicates remission and the flow cytometry results suggest cancer?
In a recent study, researchers looked at the outcomes of children with conflicting results of remission by morphology versus flow cytometry. The goal of the multi-institution effort, which included Nationwide Children’s Hospital, was to determine the frequency of discordant results and the outcomes associated with them. The results were published in the journal Leukemia.
In the vast majority of the more than 9,000 children enrolled in the Children’s Oncology Group (COG) clinical leukemia trials, morphology and flow cytometry results agreed.
But in less than 3 percent of patients, microscopic examination indicated remission while flow cytometry results showed high levels of leukemia cells.
“These children had significantly worse outcomes than children whose microscopic examination said they were remission and whose flow cytometry test confirmed those results,” says Sumit Gupta, MD, an oncologist at the Hospital for Sick Kids in Toronto, Canada, and the lead author on the recent study.
Christopher Carter, MD, a pathologist at Nationwide Children’s Hospital who was not involved in the study, says he is not surprised by the results.
“Those of us in the field who do flow cytometry have known for a long time that it is a far more sensitive, specific assay than just looking at cells under a microscope,” says Dr. Carter. “Determining the difference between an abnormal cell and a normal cell by morphological evaluation is difficult and subjective, whereas flow cytometry is an almost purely objective test.”
A recent report published in the Journal of Clinical Oncology by researchers from the United Kingdom found similar results when using a polymerase chain reaction-based technique rather than flow cytometry.
“When we put these two studies together, it supports the idea of no longer just using the microscope to define remission, but also using a molecular method like flow cytometry or PCR,” says Dr. Gupta. “The broader question is, should we jettison the microscope altogether and just use flow cytometry as our gold standard for assessing remission?”
Dr. Gupta says the next step is pooling data from groups working on this from around the world, and, if the results are the same, thinking about changing the definition of remission from microscope-based to molecular technique-based.
“Originally, there were only two central reference laboratories, at Johns Hopkins University and the University of Washington, that were appropriately equipped and had enough experience to do this assessment by flow cytometry,” says Dr. Carter.
In January 2018, Nationwide Children’s Hospital Flow Cytometry Laboratory joined the club, becoming recognized as a COG-approved lab for flow cytometry remission evaluation. It was the first laboratory in Ohio, and one of only a few children’s hospitals in the country, to receive this designation.
References:
- Gupta S, Devidas M, Loh ML, Raetz EA, Chen S, Wang C, Brown P, Carroll AJ, Heerema NA, Gastier-Foster JM, Dunsmore KP, Larsen EC, Maloney KW, Mattano LA, Winter SS, Winick NJ, Carroll WL, Hunger SP, Borowitz M, Wood BL. Flow cytometric vs morphologic assessment of remission in childhood acute lymphoblastic leukemia: A report from the Children’s Oncology Group (COG). Leukemia. 18 Dec 2018. [Accepted Article Preview]
- O’Connor D, Moorman AV, Wade R, Hancock J, Tan RMR, Bartram J, Moppett J, Schwab C, Patrick K, Harrison CJ, Hough R, Goulden N, Vora A, Samarasinghe S. Use of minimal residual disease assessment to redefine induction failure in pediatric acute lymphoblastic leukemia. Journal of Clinical Oncology. 2017;35(6):660-667.
About the author
Mary a freelance science writer and blogger based in Boston. Her favorite topics include biology, psychology, neuroscience, ecology, and animal behavior. She has a BA in Biology-Psychology with a minor in English from Skidmore College in Saratoga Springs, NY, and a PhD from Brown University, where she researched bat echolocation and bullfrog chorusing.
-
Mary Bates, PhDhttps://pediatricsnationwide.org/author/mary-bates-phd/December 27, 2016
-
Mary Bates, PhDhttps://pediatricsnationwide.org/author/mary-bates-phd/
-
Mary Bates, PhDhttps://pediatricsnationwide.org/author/mary-bates-phd/
-
Mary Bates, PhDhttps://pediatricsnationwide.org/author/mary-bates-phd/
- Post Tags:
- Cancer
- Hematology/Oncology/BMT
- Pathology
- Posted In:
- In Brief