Universal Donor CAR NK Cells: A New Platform Technology for Cancer
Universal Donor CAR NK Cells: A New Platform Technology for Cancer https://pediatricsnationwide.org/wp-content/uploads/2025/09/AdobeStock_1448558634-1024x559.jpeg 1024 559 Abbie Miller Abbie Miller https://pediatricsnationwide.org/wp-content/uploads/2023/05/051023BT016-Abbie-Crop.jpg
A first-in-human study of universal donor CAR NK cells for acute myeloid leukemia could pave a path for a new approach to treating cancer
If you ask Dean Lee, MD, PhD, the new first-in-human trial to evaluate universal donor chimeric antigen receptor (CAR) natural killer (NK) cells in patients with advanced, high-risk acute myeloid leukemia (AML) is the intersection of right time, right place and right team.
The clinical trial, which received the safe-to-proceed approval from the Food and Drug Administration (FDA) earlier this year, represents several firsts in the field:
- First CAR NK cell created on the hospital’s patented universal-donor NK cell platform
- First human trial featuring NK cells modified with the team’s patented CRISPR and AAV approach
- Four novel assays developed to ensure quality and safety
These universal donor CAR NK cells were developed and are produced completely in house at Nationwide Children’s Hospital.
“This trial represents the culmination of a lot of work over decades of my career,” says Dr. Lee, director of the Cellular Therapy and Cancer Immunotherapy program at Nationwide Children’s and The Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute. “It also represents the tremendous work of my team and colleagues. The amount of skill and tenacity required to develop new therapies based on new techniques and technologies, then to develop the assays needed to ensure safety, and finally to manage the technology commercialization and regulatory aspects to get it to a first-in-human trial is just huge, and I’m grateful to work with individuals who are so dedicated.”
Not only is the trial a milestone on its own, the research behind it establishes a new platform approach for treating cancer.
Back to the Beginning
More than 20 years ago, Dr. Lee started as a T-cell researcher making CD33 CAR T cells. When he moved to MD Anderson, he switched to NK cells after a twist of fate led him to a more effective approach to growing them in a lab — the approach that is still used today by researchers all over.
“I thought we’d just do the same things we were doing with CAR T cells to make CAR NK cells,” he says. “But it didn’t work. And eventually I moved on and focused on non-modified NK cells for about four years.”
Then, he came to Columbus, Ohio, got interested in CRISPR and hired the team for the program. The rest, they hope, is about to make history.
Universal Donor NK Cells
NK cells are the innate immune system’s first line of defense for viral infections. These white blood cells can act faster than T or B cells because they do not require prior activation or exposure to a target antigen to recognize and attack problematic cells. NK cells constantly circulate — patrolling — looking for potential tumor cells or infectious invaders.
Early in Dr. Lee’s work with NK cells, he expanded cells collected from the patient or a related donor, which were then given back to the patient after treatment to support immune function. These early studies showed that researchers can safely collect, expand and deliver NK cells to patients with cancer. But too often, the process of finding donors or collecting cells from sick kids or their family member and then expanding and delivering the cells took too long, and children grew too ill to receive the therapy.
These hurdles inspired Dr. Lee and others to question: Wouldn’t it be easier if there was an off-the-shelf product that was ready to thaw and give to patients on demand?
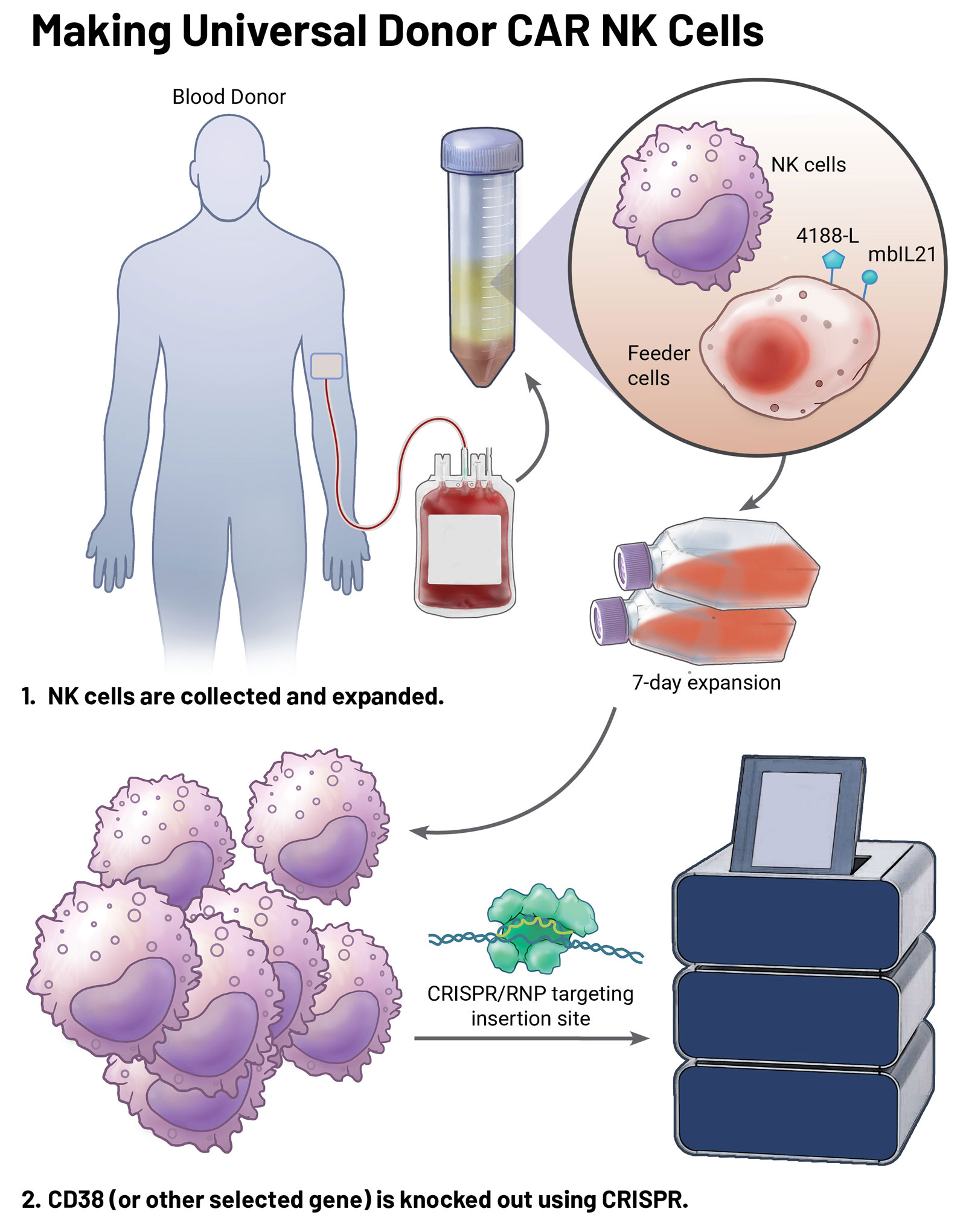
The short answer is yes. And it is what Dr. Lee has accomplished with his universal donor NK cell program. Through his research, he’s identified what makes a good donor for NK cells, and working with the National Marrow Donor Program (NMDP), formerly known as Be The Match Biotherapies, he’s developed a bank of cells that can be cryopreserved, banked and made available to patients when they need them.
According to Dr. Lee, about 1 in 20 individuals, or millions of Americans, could qualify as universal donors. Through the NMDP, donors are first identified by HLA type and KIR type. Additionally, optimal donors must have been exposed to cytomegalovirus, which leads to higher NK cell activity. Once donors are identified, a sample comes to Nationwide Children’s for a final check of the donor’s NK cell function.
After donor cells are collected at a collection center near their home, they need to be expanded. At the start, NK cells make up about 5% of the cells in the collected product from the donor. By the end of the expansion period, they make up 99%. The cells then go into cryo-storage for universal donor NK cell trials or move on to become universal donor CAR NK cells.
Working with the Nationwide Children’s Biologics Manufacturing Resource, a GMP-appropriate manufacturing facility led by Lawrence Gazda, PhD, Dr. Lee established a manufacturing process that has supported five clinical trials to date. More than 1 trillion universal donor NK cells have been delivered to patients through the program.
“The beauty of these cells is that they don’t have to match the patient to function well,” says Dr. Gazda. “And we can make these weeks, months, potentially even years ahead. I am excited to think about the potential impact for patients.”
Making CAR NK Cells
In 2017, Meisam Naeimi Kararoudi, DVM, PhD, joined Nationwide Children’s as a postdoctoral fellow. Now, he is the director of the CRISPR/Gene Editing Core and a principal investigator in the Center for Childhood Cancer Research.
“Back when I started working with Dr. Lee, everyone in the field was trying to engineer NK cells using the traditional methods used for CAR T, including lentivirus, and they were failing,” says Dr. Kararoudi. “The NK cell’s job is to get rid of viruses, and lentivirus is the same type of virus that causes HIV. We decided to use a different approach.”
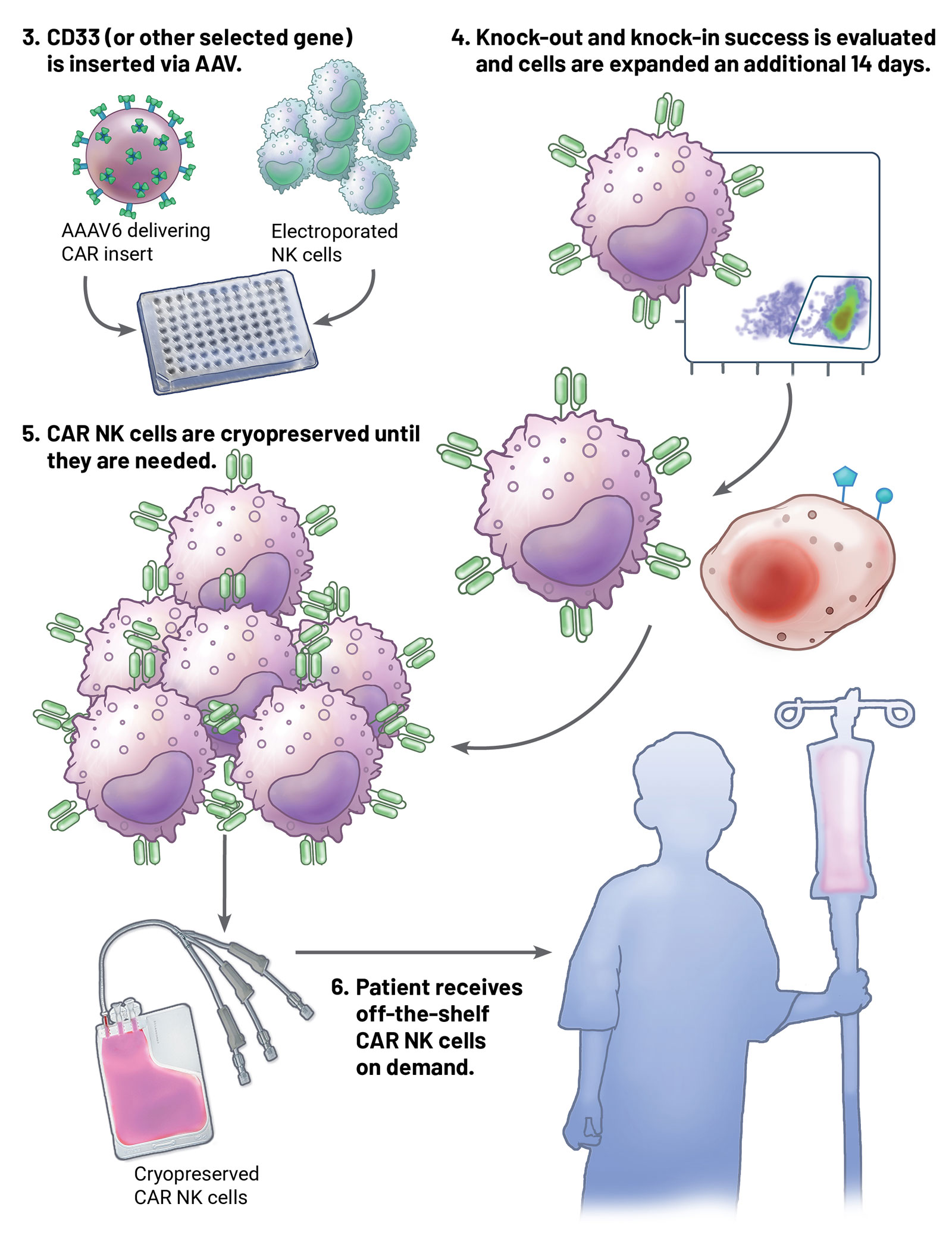
He and Dr. Lee began working together to develop a new technique to manipulate the genes in NK cells. They landed on an approach that uses CRISPR to knock out one gene, followed by using adeno-associated virus (AAV) to insert another gene in its place.
“Once we were able to knock out a gene using CRISPR, we had an open place to knock in a new gene,” says Dr. Lee. “This hadn’t been done before at the time, and it was really motivating to see it work.”
Leaning on Nationwide Children’s long history of success developing AAV-mediated gene therapies, Drs. Lee and Kararoudi worked with experts in the Jerry R. Mendell, MD, Center for Gene Therapy at Nationwide Children’s to adapt AAV systems for this approach.
The approach also solved another problem associated with using lentivirus.
“We greatly reduced the risk of random insertion into, or next to, normal healthy genes,” says Dr. Kararoudi. “Lentiviral vectors insert randomly in cells, potentially causing problems such as new cancers. The CRISPR AAV approach is site-directed, meaning the new gene only goes where we tell it to go.”
The CAR developed for the AML trial against CD33 and is inserted into the CD38 gene to make the CAR NK cell. CD33 is an important receptor found on AML cells. It is a common target of therapies treating AML, as nearly all (85-90%) AMLs express CD33.
In a paper published in Blood in 2020, the team showed that eliminating CD38 in NK cells enhanced the potency of daratumumab’s (a human monoclonal antibody targeting CD38) cytotoxic effects against multiple myeloma ex vivo.
“The novel gene editing technique used for this approach increases both safety and efficacy,” says Dr. Lee. “By knocking out the CD38, we enhance potency and potential for combined CD38 targeting. That is, any therapies targeting CD38 would not affect these CAR NK cells.”
Preclinical and Computational Studies
A key component of Dr. Lee’s research program is the preclinical lab. Marcelo De Souza Fernandes Pereira, PhD, is a research scientist in Dr. Lee’s lab.
“When it comes to the mouse experiments, I’m running everything behind the scenes,” says Dr. Pereira, who started in the Lee Lab 6 years ago as a postdoc. “These are important models that enable us to use our experimental NK cell products and see how they impact tumor cells.”
Dr. Pereira and the rest of the lab monitor the mice for decreases in tumor burden, using in vivo imaging to track tumor growth with luciferase. They also monitor for side effects such as cytokine storm, graft vs host disease and other toxicities.
“In our studies, we’ve seen very positive results using universal donor CAR NK cells,” says Dr. Pereira. “When mice are treated with CAR T cells, we often see graft vs host disease and cytokine release syndrome, also known as cytokine storm. We don’t typically see these problems in mice treated with CAR NK cells, which makes us eager to see the results of the human trial.”
In addition to studies in preclinical models, the trial is also built on data obtained from multiscale in silico modeling. In a preprint published in January in bioRxiv, Dr. Lee, Jayajit Das, PhD, and their graduate student Kun Xing use a model trained with quantitative flow cytometry and in vitro cytotoxicity data to predict the short- and long-term cytotoxicity of CD33 CAR NK cells against leukemia cell lines.
“Our model accurately predicted cytotoxicity across multiple CAR designs,” says Dr. Lee. “This adds to our evidence supporting the product currently in the clinical trial. We can also extend the model to predict CAR NK activity across many different antigens and tumor targets, giving us another tool for future applications and studies.”
Universal Donor CAR NK for AML
Margaret Lamb, MD, principal investigator of the universal donor CAR NK clinical trial and pediatric expert in the Division of Hematology, Oncology and Blood and Marrow Transplant at Nationwide Children’s, and Dr. Lee have ongoing trials testing universal donor NK cells in patients with AML. Moving the universal donor CAR NK cells forward for this patient population is a logical next step.
AML occurs in all ages of children and adults, and 50 to 80% of people with AML achieve complete remission after treatment. However, about half of those who achieve initial remission will develop relapsed AML. For those with relapsed AML (it came back after remission) or refractory (it did not respond to first-line treatments), treatment options are limited.

Margaret Lamb, MD
“In ALL [acute lymphoblastic leukemia], we have CAR T cells and other immunotherapies, which have really revolutionized the outcomes. But so far, CAR T for AML hasn’t been successful,” says Dr. Lamb. “We do not yet have an effective cell therapy product for AML. But we feel strongly that this trial could move us a step closer to changing that.”
The lack of a CAR T option for AML creates a prime opportunity to use the universal donor CAR NK cell platform.
Drs. Lee and Lamb have several reasons for thinking that CAR NK cells may be better than CAR T cells for AML, all supported by previous studies of NK cells in AML and preclinical studies of the universal donor CAR NK cells:
- First, CAR NK cells and CAR T cells have similar effector functions, but the NK cell activating receptors plus the CAR-antigen recognition may enhance potency and minimize antigen escape.
- Additionally, using healthy donor NK cells does not carry the risk of graft vs host disease like donor T cells.
- Collecting cells from patients with refractory AML is challenging. AML cells can repress the function of the patients’ own T cells, making them poor candidates for T cell collection and modification for an autologous CAR T product.
- Using healthy donor cells for the CAR NK therapy means that the product contains more fit and functional cells than those collected from sick patients.
- Cytokine reactions are significant concerns for CAR T cell therapy. However, these strong reactions have not been observed to the same degree in CAR NK cell studies.
“One reason we’re so optimistic about the potential for CAR NK cells for AML is the low rates of cytokine storm produced by NK cell therapies,” says Dr. Lamb. “We think this will make them better suited to AML.”
Ensuring Safety, One Innovation at a Time
Dr. Lee’s team has innovated every step of the process to develop the CAR NK cell therapy for AML and bring it to this point — even developing quality assurance tests.
One of the challenges with novel therapeutics is the need for new assays and tests to ensure their safety. The team needed to be able to confidently say that the product contained only the intended cells with the intended genetic alterations with the intended purity and concentration. Not only are these tests needed for the research process, but they also are essential to the regulatory processes required for moving discovery and innovation into the clinic.
Cecele Denman, MS, began working with Dr. Lee a decade ago at MD Anderson. At Nationwide Children’s, she is the laboratory manager for the Flow Cytometry and Immune Monitoring Cores, which provide support for Dr. Lee’s work. She developed the novel autonomous outgrowth assay that ensures that the NK cells haven’t acquired any cancer potential before delivery to patients. This test is part of the testing required by the FDA.
NK Cell Trials at Nationwide Children’s and The Ohio State University
- Studies involving universal donor TGF-beta imprinted NK cells for sarcoma, neuroblastoma, brain tumors, breast, melanoma, multiple myeloma and T-cell L/Ly.
- Universal donor expanded NK cells for relapsed/refractory AML in children and adults.
- Haploidentical expanded NK cells for AML.
- More than 1 trillion universal donor cells delivered to participants in clinical trials.
Yasemin Sezgin, MS, PhD, is a research associate and lab manager in the Kararoudi Lab. She also developed assays for FDA and regulatory compliance. She developed a novel assay to ensure that the AAV has not become active in the cells, and that the cells have the correct number of copies of the transgene in the final product. In addition, she also helps interpret the test for on and off target effects that looks for chromosomal abnormalities after gene editing. The team developed this test in collaboration with the Institute for Genomic
Medicine. Lastly, the team worked with KROMATID, Inc. and Cergentis, B.V. to develop assays that ensure that the CRISPR/AAV process inserted only the CD33 CAR and only in the correct place.
“We want to make sure that the final product has enough, but not too many, copies of the inserted gene in the cells,” says Dr. Sezgin.
“Ensuring cell therapy products are safe, reproducible and high-quality is our focus,” adds Denman. “It’s how we promote the best possible outcomes for children and contribute to improved therapies for kids with cancer.”
Enrolling Now
The non-randomized, open-label, dose-escalation study, began enrolling participants in August 2025.
Key inclusion criteria for the study include relapsed or primary refractory CD33+ AML including second or subsequent relapse or any relapse after hematopoietic stem cell transplant. Participants must have a HSCT donor identified, as the goal is to get the participants into remission and ready for transplant.
“CD33 is also present on normal myeloid cells,” says Dr. Lamb. “We need to find out if this therapy increases risk for aplasia [failure produce blood cells]. This is one reason we need to have a HSCT donor identified for participants.”
In addition to receiving the universal donor CAR NK cells, participants in the trial will receive the AML-directed reinduction regimen with venetoclax. Previous research shows that adding venetoclax enhances NK cell function, according to Dr. Lamb.
“This is a pediatric study, but because it is first-in-human, our first three patients at each dose escalation will need to be 16 years old or older,” says Dr. Lamb. “After those initial doses, we look forward to including participants of all ages.”
While this is currently a single-site study, Drs. Lee and Lamb both hope that a multisite opportunity will grow from this study.
“The off the shelf product and large-scale manufacturing makes a multi-institutional study feasible and could change the way we approach cell therapy,” says Dr. Lamb.
Future Forward
The universal donor approach as a platform technology enables the development of off -the-shelf CAR NK cells targeting many different cancers. Th e team says they have developed more than a dozen different CARs targeting unique cancer antigens, and they recently received a large grant to pursue CAR NK cells for osteosarcoma.
“AML is just the beginning,” says Dr. Lee. “It is an exciting time to work with CAR NK cells, and our universal donor program opens a lot of doors for multicenter collaboration. I can’t wait to see what happens next.”
“The off -the-shelf CAR NK cell product is truly revolutionary in that it supports an incredibly fast turnaround time for patients to be treated,” adds Dr. Lamb. “We can go from consenting a patient for participation to infusion in a single day. That has the potential to change outcomes.”
This article appeared in the 2025 Fall/Winter print issue. Download the issue here.
References:
- Ahmad S, Xing K, Rajakaruna H, Stewart WC, Beckwith KA, Nayak I, Kararoudi MN, Lee DA, Das J. A framework integrating multiscale in-silico modeling and experimental data predicts CD33CAR-NK cytotoxicity across target cell types. bioRxiv. 2025 Jan 2:2024.12.31.630941.
- Ciurea SO, Kongtim P, Srour S, Chen J, Soebbing D, Shpall E, Rezvani K, Nakkula R, Thakkar A, Troy EC, Cash AA, Behbehani G, Cao K, Schafer J, Champlin RE, Lee DA. Results of a phase I trial with Haploidentical mbIL-21 ex vivo expanded NK cells for patients with multiply relapsed and refractory AML. American Journal of Hematology. 2024;99(5):890-899.
- Denman CJ, Senyukov VV, Somanchi SS, Phatarpekar PV, Kopp LM, Johnson JL, Singh H, Hurton L, Maiti SN, Huls MH, Champlin RE, Cooper LJ, Lee DA. Membrane-bound IL-21 promotes sustained ex vivo proliferation of human natural killer cells. PloS One. 2012;7(1):e30264.
- Dutour A, Marin V, Pizzitola I, Valsesia-Wittmann S, Lee D, Yvon E, Finney H, Lawson A, Brenner M, Biondi A, Biagi E, Rousseau R. In vitro and in vivo antitumor effect of anti-CD33 chimeric receptor-expressing EBV-CTL against CD33 acute myeloid leukemia. Advances in Hematology. 2012;2012:683065.
- Lee DA. Cellular therapy: Adoptive immunotherapy with expanded natural killer cells. Immunology Reviews. 2019;290(1):85-99.
- Molica M, Perrone S, Mazzone C, Niscola P, Cesini L, Abruzzese E, de Fabritiis P. CD33 Expression and gentuzumab ozogamicin in acute myeloid leukemia: Two sides of the same coin. Cancers (Basel). 2021 Jun 28;13(13):3214.
- Naeimi Kararoudi M, Nagai Y, Elmas E, de Souza Fernandes Pereira M, Ali SA, Imus PH, Wethington D, Borrello IM, Lee DA, Ghiaur G. CD38 deletion of human primary NK cells eliminates daratumumab-induced fratricide and boosts their effector activity. Blood. 2020;136(21):2416-2427.
- Romain G, Senyukov V, Rey-Villamizar N, Merouane A, Kelton W, Liadi I, Mahendra A, Charab W, Georgiou G, Roysam B, Lee DA, Varadarajan N. Antibody Fc engineering improves frequency and promotes kinetic boosting of serial killing mediated by NK cells. Blood. 2014;124(22):3241-3249.
- Silla L, Valim V, Pezzi A, da Silva M, Wilke I, Nobrega J, Vargas A, Amorin B, Correa B, Zambonato B, Scherer F, Merzoni J, Sekine L, Huls H, Cooper LJ, Paz A, Lee DA. Adoptive immunotherapy with double-bright (CD56bright/CD16bright) expanded natural killer cells in patients with relapsed or refractory acute myeloid leukaemia: A proof-of-concept study. British Journal of Haematology. 2021;195(5):710-721.
Image credit: Adobe Stock (header); Nationwide Children’s (portraits and illustration)
About the author
Abbie (Roth) Miller, MS, MWC, is a passionate communicator of science. As the manager of medical and science content at Nationwide Children’s Hospital, she shares stories about innovative research and discovery with audiences ranging from parents to preeminent researchers and leaders. She is a Medical Writer Certified®, credentialed by the American Medical Writers Association, and received her masters of science in Health Communication from Boston University.
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/