Targeting DIPG: The Most Puzzling of Pediatric Brain Tumors
Targeting DIPG: The Most Puzzling of Pediatric Brain Tumors https://pediatricsnationwide.org/wp-content/uploads/2021/04/Cover-Final-Color-V4-Flat-RGB-web-crop-2.jpg 772 447 Katie Brind'Amour, PhD, MS, CHES Katie Brind'Amour, PhD, MS, CHES https://pediatricsnationwide.org/wp-content/uploads/2021/03/Katie-B-portrait.gif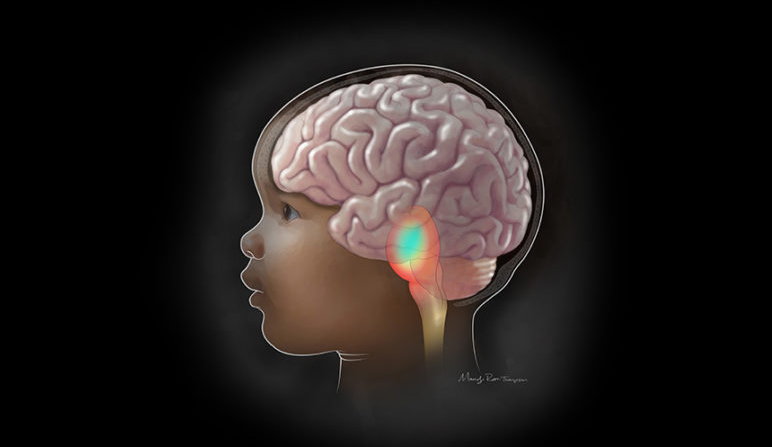
Survival has dramatically improved for numerous pediatric cancers over the last several decades, with a notable and very deadly exception: diffuse intrinsic pontine glioma (DIPG). Now, a community of researchers and clinician-scientists have set the stage for a renewed — and better-armed — assault against this beast of a brain tumor.
The past 50 years have brought story after story of success in the field of pediatric oncology. In 1975, acute lymphoblastic leukemia had a 5-year survival rate of 57%; by 2012, it reached 92%.
Non-Hodgkin lymphoma followed a similar trajectory, with a 5-year survival rate of only 43% in 1975, reaching 91% by 2012.
And childhood cancer in general became much less deadly during this time period, with an overall 5-year survival of 58% in the 1970s surpassing 83% by 2014.
As the most common pediatric cancer, childhood leukemia, became increasingly curable, oncologists were left with a new main foe: brain cancers, which have supplanted leukemia as the leading cause of childhood cancer deaths.
Yet even in certain pediatric brain tumors, progress has been tremendous. Oligodendroglioma and pilocytic astrocytoma both have 5-year survival rates of 90% or greater.
Medulloblastoma — a historically challenging tumor despite decades of creative combination therapy studies — has offered great hope to neurooncologists and cancer biologists, as increased understanding of molecular subgrouping and targeted therapies have raised survival for certain subgroups to 80-90%.
Despite these steady and substantial improvements, diffuse intrinsic pontine glioma (DIPG) survival remains dismal: median survival is only 8-11 months, with 5-year survival at just 2%.
In the past half-century of oncology care and research, pediatric cancer patients have benefited from skyrocketing survival rates for many diagnoses, with an overall 5-year survival of 83%. Not so for DIPG, for which nearly all efforts have failed: only 2% of children survive 5 years, and more than half are dead in less than a year.
While other cancers benefitted from targeted therapies and advances in tumor resection techniques, DIPG tumors have remained virtually untouchable despite inclusion in more than 250 clinical trials for chemotherapies, targeted therapy, combination therapies and immunotherapies. Although some patients respond briefly to radiation (which may extend life about 3 months), the cancer virtually always returns quickly, and to deadly effect.
The tumor is rare, with only 200-300 cases diagnosed each year in children in the United States. Its location in the brain’s pons — the control center, directing the body’s most basic and essential functions — makes DIPG hard to access for biopsy or drug delivery. The diffuse nature of the tumor also makes it essentially unresectable; tumor cells cannot be removed without taking vital healthy tissue with them.
These characteristics have made DIPG both difficult to treat and difficult to learn about. As the advent of molecular profiling and cancer genomics catapulted numerous cancer types into the realm of targeted therapies and increased cure rates, DIPG tissue samples to study were in short supply.
“It’s a rare disease, but it’s killing people — it’s like a death sentence,” says Rachid Drissi, PhD, who recently joined the Center for Childhood Cancer and Blood Diseases in the Abigail Wexner Research Institute at Nationwide Children’s Hospital as a principal investigator. He has dedicated his career to studying the biology of high-risk pediatric brain tumors. “That’s where, as a basic scientist, I became very interested in DIPG. If it is incurable, that means we don’t know the biology. Otherwise, we would find the weakness and target that weakness. So the question we all need to answer is simple: What is the biology of DIPG?”

DIPG scientists and clinicians have faced an uphill battle.
But dedicated experts from around the world — together with the families and patient advocacy organizations providing financial and practical support to make this work possible — are methodically tackling each hurdle in turn. And as they set the stage for more rapid advancements against this deadly disease, members of the DIPG research community are becoming increasingly optimistic about what the next 5-10 years will hold.
Getting Past Location
After years facing down DIPG’s disappointing track record in clinical trials, researchers have pinpointed one likely physical reason as the blood-brain barrier: a cellular filtration system that prevents systemic therapies from crossing into the brain.
Many experts now believe that the majority of chemotherapy and other treatments delivered historically via standard methods to children with DIPG didn’t reach the tumors at all, or did so in such small quantities as to be rendered ineffective. To get around this natural barrier, clinician-scientists have been busy exploring and fine-tuning novel delivery methods, such as focused ultrasound technology (which may temporarily disrupt the blood-brain barrier to allow the drugs to pass into the brain), nanoparticles and direct drug delivery of chemotherapy into the tumor.
“If we deliver drugs directly into target tissue, we will achieve higher concentrations than conceivable via the systemic route, with less toxicity,” says Mark Souweidane, MD, director of Pediatric Neurosurgery, New York-Presbyterian/Weill Cornell Medical Center and Memorial Sloan Kettering Cancer Center.
Dr. Souweidane has been working nearly two decades to operationalize the mechanical and technical aspects of chemotherapy infusion directly into brain tumors through a small catheter or cannula. His work with DIPG patients has demonstrated that direct injection can enable more than 1000-times the drug concentration in the target tissue than anywhere in systemic circulation, with no appreciable systemic toxicity.
“That has been a huge leap forward,” says Dr. Souweidane, who published his Phase I findings in The Lancet: Oncology in 2018. “Now the challenge is how to optimize this approach to make a positive clinical impact on this disease.”
In addition, basic scientists now also have the key resource at their disposal that, for decades, was denied them: tissue samples.
For many years, biopsies of DIPG were not performed. The location of the tumor in the pons left many clinicians and families fearing that any misstep during the procedure could result in significant morbidity — or even death. As a result, researchers had only limited amounts of postmortem tissue to study, which may have mutated considerably after diagnosis due to treatments or tumor evolution.
“The biggest thing hindering research progress was the lack of tumor tissue,” says Chris Jones, PhD, head of the Glioma Team and full professor of Childhood Brain Tumor Biology at The Institute of Cancer Research in London. “Families on both sides of the Atlantic pushed for safe biopsy procedures to provide labs like mine with the tissue we needed to study DIPG, so the next generation of kids hopefully won’t have to go through what they did. They came together and catalyzed the last 15 years of DIPG research.”
The result was transformative. In 2012, researchers including those now leading the Steve and Cindy Rasmussen Institute for Genomic Medicine (IGM) at Nationwide Children’s, profiled the genome of seven DIPG tumors (and then performed targeted screening in an additional 43 tumors) and uncovered an abnormality common to nearly four in every five cases: a mutation called K27M in the histone H3 family of genes. Previously thought of as an untouchable portion of the genome in terms of mutations, the group of histone H3 variants were unique to human biology at the time.
That’s when the clock started ticking toward a biology based cure for DIPG.
A NEW ERA FOR DIPG BIOLOGY
With the seminal genomic study and almost a decade of research to follow, scientists now have preliminary genome-based classifications for DIPG tumors. The work has shown, however, that numerous mutations — sometimes within a single tumor — may all be materially involved in the disease and its progression.
“We now know there is important heterogeneity across patients, in that no two patients have the same list of mutations as one another. And there is heterogeneity within an individual’s DIPG tumor as well — mutations occur at different stages and in different cells, creating a complicated mosaic of tumor subclones with different mutations interacting with each other,” says Professor Jones, who led a multi-institutional effort to identify key mutations in 322 DIPG tumors (and more than 600 other high-grade gliomas), published in Cancer Cell in 2017. The effort identified numerous additional genomic subgroups for DIPG and examined prognosis related to the mutations.
“It’s a complicated profile,” Professor Jones says, “and genomics helps unravel the details and may guide treatments we might give to patients with this disease.”
Histone tail involvement points to a role for the cellular microenvironment and epigenetics in the development of DIPG. The histone tail interacts with the rest of a cell’s DNA strand, signaling to turn certain genes on or off. Unfortunately, K27M mutations remain elusive targets from a therapeutic standpoint; new drugs must precisely impact the histone tail so that it sends proper signals for gene expression.
Utilizing other information revealed by the genomics studies, Professor Jones and his research team are developing what may be the first drug designed to specifically target a childhood brain tumor mutation, expected to enter clinical trials in the coming year or two.
His compound targets somatic mutations in ACVR1, which are present in about 25% of DIPG cases. Additional efforts aim to connect clinical practice with existing genomic information to allow refinement of the current subgroups and prognostic data.
“We wanted to see if there was a correlation between tumor features seen in serial clinical imaging and those actual tumors’ genomics and expression of genes,” says Dr. Drissi.
He and his team have explored this concept of “radiogenomics” in DIPG and successfully identified imaging characteristics for certain prognoses and DIPG subgroups, such as those with H3F3A mutations (who have a poor response to radiation therapy) or those with numerous mutations and inflammatory tumor profiles (who have a favorable initial response radiographically to radiation). They published the work this year in Acta Neuropathologica Communications.
Radiogenomics may be imminently helpful in the clinic, as imaging is the first thing done when a neurological abnormality is suspected. For example, H3F3A cases could potentially benefit from upfront craniospinal irradiation, Dr. Drissi suggests, while those with initial responses to radiation could potentially benefit from post-radiation immunotherapy.
“Time is of the essence for these patients,” says Dr. Drissi. “The ultimate goal is that from initial imaging, we can tell what each patient’s subtype is, the best treatment options and what their prognosis will be.”
Dr. Drissi’s continued work in genomics-informed DIPG research will now involve the IGM team at Nationwide Children’s, which offers genomic profiling for research purposes and for patients with recurrent, treatment-resistant or rare tumors. Theoretically, radiogenomic data could also be paired with knowledge obtained from another emerging research tool: “liquid biopsies,” or samples of cerebrospinal fluid and blood taken periodically post-diagnosis.
“We need to make sure that any child with DIPG entering clinical trials contributes tissue and other samples in order to understand the changes in the tumor and its environment across time and treatment,” says Professor Jones, who is also the preclinical lead for the CONNECT Consortium, an international research collaborative focused on DIPG and other high-risk pediatric brain cancers. “We need to understand the basic biology of the tumor itself in order to develop rational, smart combination therapies, test them in preclinical models and collectively figure out what’s working, so we can get it to the next phase more rapidly.”
Genomics profiling work and science-based clinical studies have also prompted tumor microenvironment studies of DIPG. Together they may further explain why the cancer has not fallen prey to natural immune system defenses or immunotherapies tried to date.
DIPG is considered an immunologically “cold” tumor in that it incites very little, if any, immune response or inflammation, likely because it results from relatively few mutations (compared to adult cancers) and has no obvious antigen that tells the immune system to target it.
“We are in the process of understanding the tumors’ strategies for hiding from the immune system,” Dr. Drissi says. “Once we get to know how the tumor avoids it, our job will be to push DIPG cells to express genes to make them look like invaders, to tell the body, ‘You need to attack these cells!’”
These unique and varied approaches all attempt to exploit the biology of the tumor as information accrues from tissue samples, novel models and genomics studies. Just having access to this robust information that enables thorough biological examination — for the first time since the cancer’s discovery — means research is able to progress on many fronts.
ACCELERATING TRANSLATIONAL RESEARCH
“In recent years, basic science research in DIPG has provided new and exciting insights in the cell-intrinsic vulnerabilities of DIPG cancer cells, the interactions with healthy brain cells that drive the growth of DIPG and immuno-therapeutic opportunities for DIPG,” says Michelle Monje Deisseroth, MD, PhD, associate professor of Neurology and Neurological Sciences at Stanford University. “Translating those discoveries towards effective therapies for children with DIPG in the next 5-10 years will require cooperation and collaboration among academic children’s hospitals, like those that participate in pediatric brain tumor clinical trials consortia.”
To this end, Maryam Fouladi, MD, pediatric neurooncologist and the new co-executive director of the Pediatric Neuro-Oncology Program at Nationwide Children’s, founded the CONNECT Consortium in 2012. The international collaborative of pediatric cancer research and clinical care centers exists to improve outcomes for children with high-risk brain tumors such as DIPG and other high-grade gliomas, and will now be based out of Nationwide Children’s.
“DIPG is a wily tumor,” says Dr. Fouladi, who is currently leading a trial to test efficacy and safety of combining a BMI-1 inhibitor with radiation therapy in children with DIPG and other high-grade gliomas (CONNECT1702); the study is based on findings from basic science collaborations with Dr. Drissi and others, described in a 2020 publication in Molecular Cancer Research. “It develops resistance and survives through multiple different pathways, so we will need a multi-pronged approach to trying to cure it.”
The 18 member centers of the CONNECT Consortium examine (in their own labs and models) promising therapies emerging from preclinical and Phase I studies, hoping to identify the most fruitful potential drug combinations for DIPG. Provided positive early results are replicable, CONNECT members rapidly design scientifically rational pilot studies to evaluate these combination therapies in the clinic — often with seed money made available by the companies that developed the drugs — to gather preliminary safety and efficacy data. Then they share the group’s findings with larger consortia, such as the Children’s Oncology Group, so that tolerable and auspicious combination therapies can be rapidly moved into more mainstream Phase II/III testing.
“We want to build on what we know works right now — radiation — not just throw every method at these children and adversely affect their quality of life,” says Dr. Fouladi. “We want to judiciously, carefully conduct clinical trials looking at both toxicity and efficacy, combining radiation with chemotherapy, immunotherapy or targeted therapy. Progress will be incremental, but in the end, all of these are likely to become part of an effective armamentarium against DIPG in some way.”
Greater understanding of biology and scientifically sound drug combinations revealed by consortium-based studies have also aided further study of clinically viable tools for drug delivery.
Dr. Souweidane has a multi-center Phase II clinical trial in development to examine the capacity of the highly technical platform of direct chemotherapy injection to reach a far greater community. Anecdotally, direct chemotherapy injection has already yielded some encouraging results, with a handful of patients from Dr. Souweidane’s Phase I trial surviving beyond 3 years (median survival was 15.3 months) and another two patients considered cured.
“We’ve taken care of the technical, mechanical and surgical hurdles, so the next step is to demonstrate clinical benefit: optimizing direct chemotherapy injection for enhanced distribution of the drug, maximizing tumor coverage, defining tumor response criteria, adding rounds of treatment and building on the solid conceptual foundation of the platform,” says Dr. Souweidane. “If we can control the tumor at its major point of origin, then supplement with multimodal therapy — systemic treatment, external beam radiation or another modality to help avoid distant recurrence — I have no misgivings about what we can accomplish with this.”
With thoughts of multimodal care in mind, Dr. Fouladi will collaborate with The Ohio State University Wexner Medical Center as the two institutions open central Ohio’s first proton therapy center for targeted radiotherapy in 2021. Together with colleagues in radiation oncology at the university, Dr. Fouladi hopes to explore numerous additional ways to optimize radiation for children by minimizing dosage and toxicity.
Coupled with the efforts from the DIPG Registry — also founded and chaired by Dr. Fouladi to advance DIPG research via the collection and sharing of tissue samples — and other, larger pediatric cancer consortia (such as the Pediatric Brain Tumor Consortium, the Pacific Pediatric Neuro-Oncology Consortium and the NEXT Consortium, all of which now claim Nationwide Children’s as a member or host institution), things are looking up.
The DIPG Registry, for example, includes more than 1200 patients at about 115 sites in 15 countries, pooling clinical imaging, pathology findings and tissue samples to enable large-scale studies of DIPG biology. The registry has enabled 140 disease models and more than 30 collaborative studies, with numerous publications to share valuable data with the broader scientific community. The registry’s genomics efforts will now be based out of Nationwide Children’s IGM.
“Being part of multiple large, organized consortia is really a major privilege, as it opens the door to our patients to be part of clinical trials and receive novel treatments that wouldn’t be available otherwise,” says Ralph Salloum, MD, another recent addition to the neuro-oncologist team at Nationwide Children’s; he is a principal investigator for the Pediatric Brain Tumor Consortium and study chair of two studies through the consortium. “DIPG tumors are rare. These consortia allow our trials to open to patients at other institutions, so that they get access to the trial and we get enough patients to answer research questions and advance the field — it’s a mutually beneficial relationship.”
IMPROVING SURVIVAL, AS FAST AS POSSIBLE
For decades, DIPG has been the biggest challenge in the world of pediatric cancer. But with biology and genomics data, tissue samples, consortia, funding opportunities and new delivery techniques falling into place, experts around the world believe they are well positioned to make major inroads in DIPG care and survival.

“This is not a disease anyone can cure on their own — it will take more than just neurosurgeons, just clinicians, just geneticists or just pharmacokineticists — it’s truly going to take a village,” says Dr. Fouladi. “The beauty of the DIPG community is that patients and parents are unbelievably supportive and the researchers and clinicians are extremely collaborative. By being so, we’ve made huge strides together. Now we have to take all of this understanding of biology and figure out how to turn it into a cure.”
These combined advances are likely to result in incremental progress, through which a true “cure” is really more of a steady crawl toward improved survival.
“There is a desperation we all feel, but I can’t say we’re going to cure it in the next 5 years — a whole lot still needs to be done,” says Jeffrey Leonard, MD, co-executive director of the Pediatric Neuro-Oncology Program and chief of Neurosurgery at Nationwide Children’s. His research focuses on the genetics of pediatric brain tumors and potential immunomodulatory and exosome therapies. “There needs to be a significant advancement in our understanding of DIPG tumor pathogenesis, and we are grateful to be one of the centers leading that.”
As advancements roll in, neuro-oncologists may eventually be able to shift some of their efforts to other positives that come with climbing survival statistics: what happens after DIPG treatment success, when patients finally have a future ahead of them.
When that time arrives, Dr. Fouladi and Dr. Salloum will have additional fuel for their research, which has long included the reduction of post-therapy late effects of treatment, often via a better understanding of tumor biology, improved treatment techniques or the reduction in dosage required to shrink tumors. Dr. Salloum joined Nationwide Children’s in part to spearhead the development of a new multidisciplinary brain tumor survivorship program.
The prospect of survivors has been elusive to date, but it is an exciting one for those who have devoted their careers to DIPG research.
“If we can find a cure for this, it would be huge, because if you asked what cancer has the worst prognosis in pediatrics, most people would tell you it is DIPG,” says Dr. Fouladi. “One day I hope to say it isn’t. That’s why we need to focus on it. We have a lot of great people working together now — without any egos and for the right reasons — to get this done.”
This article is found in the Spring/Summer 2021 print issue. Download the full issue.
References:
- Aziz-Bose R, Monje M. Diffuse intrinsic pontine glioma: molecular landscape and emerging therapeutic
targets. Current Opinion in Oncology. 2019 Nov;31(6):522-530. - Mackay A, Burford A, Carvalho D, Izquierdo E, Fazal-Salom J, Taylor KR, Bjerke L, Clarke M, Vinci M, Nandhabalan M, Temelso S, Popov S, Molinari V, Raman P, Waanders AJ, Han HJ, Gupta S, Marshall L, Zacharoulis S, Vaidya S, Mandeville HC, Bridges LR, Martin AJ, Al-Sarraj S, Chandler C, Ng HK, Li X, Mu K, Trabelsi S, Brahim DH, Kisljakov AN, Konovalov DM, Moore AS, Carcaboso AM, Sunol M, de Torres C, Cruz O, Mora J, Shats LI, Stavale JN, Bidinotto LT, Reis RM, Entz-Werle N, Farrell M, Cryan J, Crimmins D, Caird J, Pears J, Monje M, Debily MA, Castel D, Grill J, Hawkins C, Nikbakht H, Jabado N, Baker SJ, Pfister SM, Jones DTW, Fouladi M, von Bueren AO, Baudis M, Resnick A, Jones C. Integrated molecular meta-analysis of 1,000 pediatric high-grade and diffuse intrinsic pontine glioma. Cancer Cell. 2017 Oct 9;32(4):520-537.e5.
- Senthil Kumar S, Sengupta S, Zhu X, Mishra DK, Phoenix T, Dyer L, Fuller C, Stevenson CB, DeWire M, Fouladi M, Drissi R. Diffuse intrinsic pontine glioma cells are vulnerable to mitotic abnormalities associated with BMI-1 modulation. Molecular Cancer Research. 2020 Nov;18(11):1711-1723.
- Souweidane MM, Kramer K, Pandit-Taskar N, Zhou Z, Haque S, Zanzonico P, Carrasquillo JA, Lyashchenko SK, Thakur SB, Donzelli M, Turner RS, Lewis JS, Cheung NV, Larson SM, Dunkel IJ. Convection-enhanced delivery for diffuse intrinsic pontine glioma: a single-centre, dose-escalation, phase 1 trial. Lancet Oncology. 2018 Aug;19(8):1040-1050.
- Wu G, Broniscer A, McEachron TA, Lu C, Paugh BS, Becksfort J, Qu C, Ding L, Huether R, Parker M, Zhang J, Gajjar A, Dyer MA, Mullighan CG, Gilbertson RJ, Mardis ER, Wilson RK, Downing JR, Ellison DW, Zhang J, Baker SJ; St. Jude Children’s Research Hospital–Washington University Pediatric Cancer Genome Project. Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nature Genetics. 2012 Jan 29;44(3):251-253.
- Zhu X, Lazow MA, Schafer A, Bartlett A, Senthil Kumar S, Kumar Mishra D, Dexheimer P, DeWire M, Fuller C, Leach JL, Fouladi M, Drissi R. A pilot radiogenomic study of DIPG reveals distinct subgroups with unique clinical trajectories and therapeutic targets. Acta Neuropathologica Communications. 2021;9(14).
Image credits: Nationwide Children’s
About the author
Katherine (Katie) Brind’Amour is a freelance medical and health science writer based in Pennsylvania. She has written about nearly every therapeutic area for patients, doctors and the general public. Dr. Brind’Amour specializes in health literacy and patient education. She completed her BS and MS degrees in Biology at Arizona State University and her PhD in Health Services Management and Policy at The Ohio State University. She is a Certified Health Education Specialist and is interested in health promotion via health programs and the communication of medical information.
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 28, 2014