Brain Organoids Provide Novel Mechanistic Insight into Microcephaly Associated with AUTS2 Syndrome
Brain Organoids Provide Novel Mechanistic Insight into Microcephaly Associated with AUTS2 Syndrome https://pediatricsnationwide.org/wp-content/uploads/2022/08/Hester_cerebral-organoid-pic_2-WEB.jpg 873 552 Lauren Dembeck Lauren Dembeck https://pediatricsnationwide.org/wp-content/uploads/2021/03/Dembeck_headshot.gif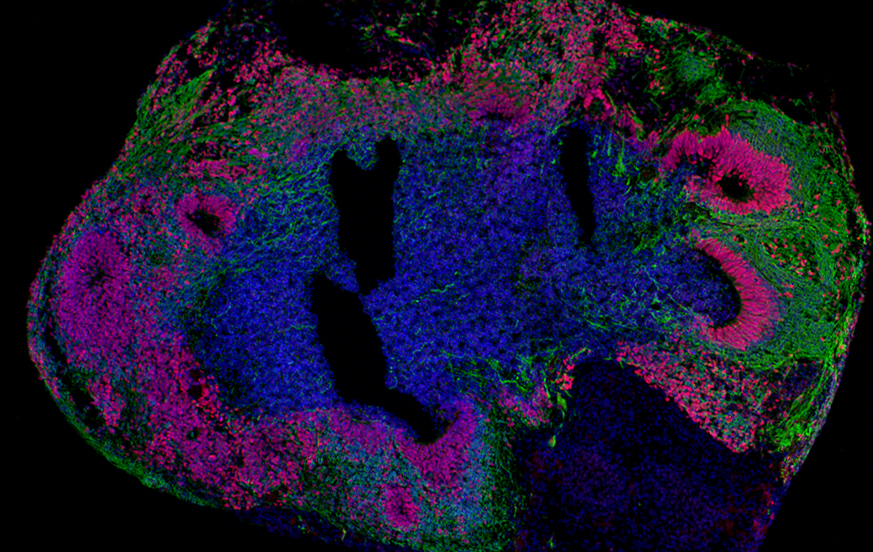
Researchers have functionally validated a genotype-phenotype relationship between a variant in the AUTS2 gene and neurodevelopmental deficits in AUTS2 syndrome using patient-derived brain organoids.
Genetic variants in more than 100 genes, including the gene AUTS2, have been associated with autism spectrum disorders. However, the model systems used to study the mechanisms underlying how these genetic variants cause these neurodevelopmental disorders often do not adequately recapitulate disease phenotypes.
For the first time, researchers at Nationwide Children’s Hospital have established a brain organoid model of AUTS2 syndrome using patient-derived cells and confirmed a causal relationship between a genetic variant in the AUTS2 gene and neurodevelopmental deficits in AUTS2 syndrome. The study was published in Brain.
“We are working very closely with clinicians to help better diagnose patients using genomics,” says senior study author Mark Hester, PhD, who is a principal investigator in the Steve and Cindy Rasmussen Institute for Genomic Medicine at Nationwide Children’s. “Our clinical genetics team referred us to a patient who presented with several neurodevelopmental conditions, such as microcephaly, epilepsy, profound intellectual disability, as well as some dysmorphic features. Multiple clinical tests performed on the patient were unable to yield a diagnosis.”
The Nationwide Children’s genomics team performed whole genome sequencing on the patient and identified a variant in AUTS2. Other variants in AUTS2 have been associated with a broad spectrum of neurological conditions and congenital brain malformations. Using bioinformatics tools, the team predicted the patient’s AUTS2 variant was pathogenic. However, to confirm that a variant is causing a phenotype, functional validation studies of the variant are necessary.
“In AUTS2 syndrome, patients often present with intellectual disability and microcephaly,” explains Dr. Hester, who also holds appointments at The Ohio State University. “We hypothesized that brain organoids could recapitulate the microcephalic phenotype and provide mechanistic insight into the disorder.”
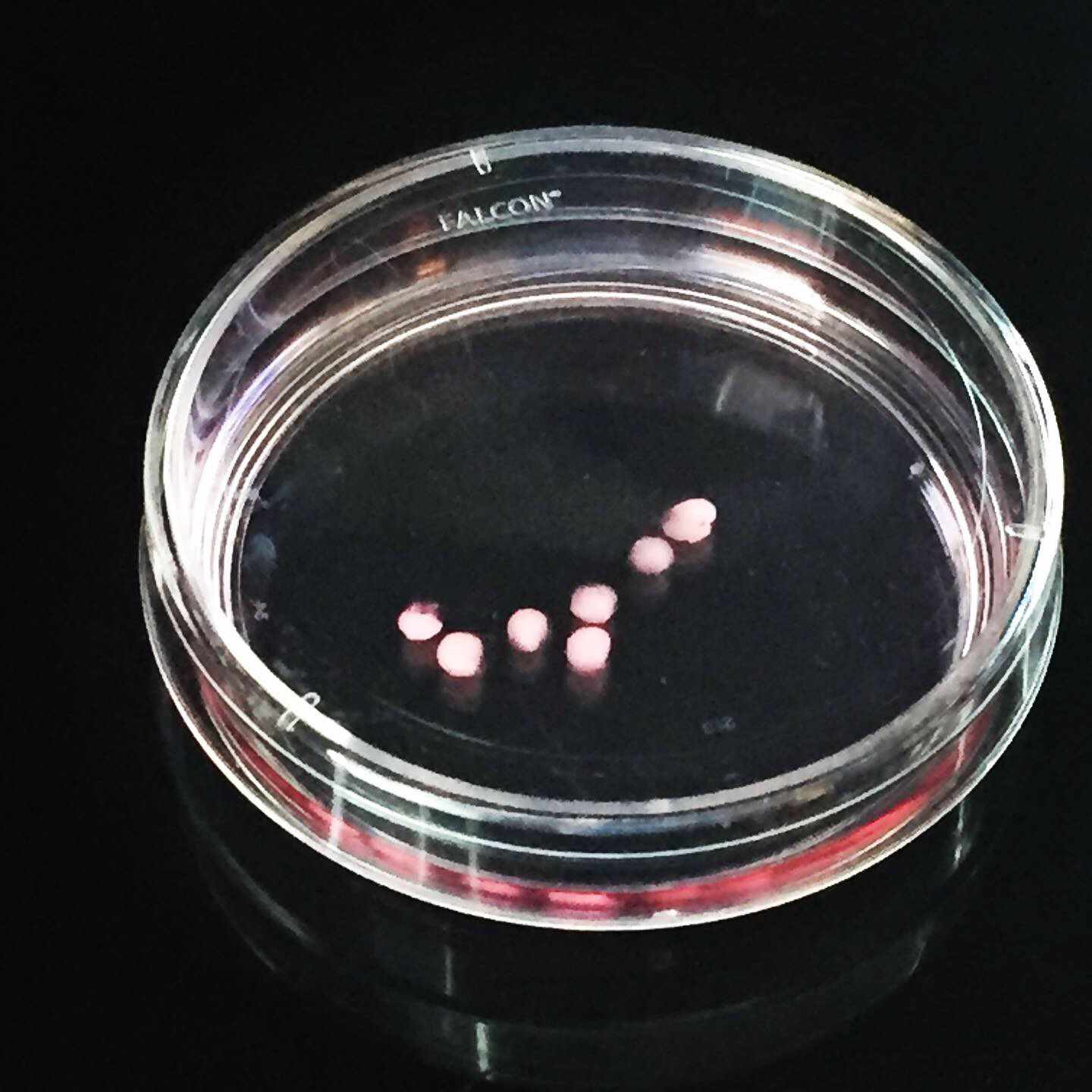
To investigate the pathophysiology of the patient’s AUTS2 variant, Dr. Hester and his team used patient-derived induced pluripotent stem cells to create a brain organoid model, self-organizing three-dimensional structures recapitulating complex developmental features of the human brain.
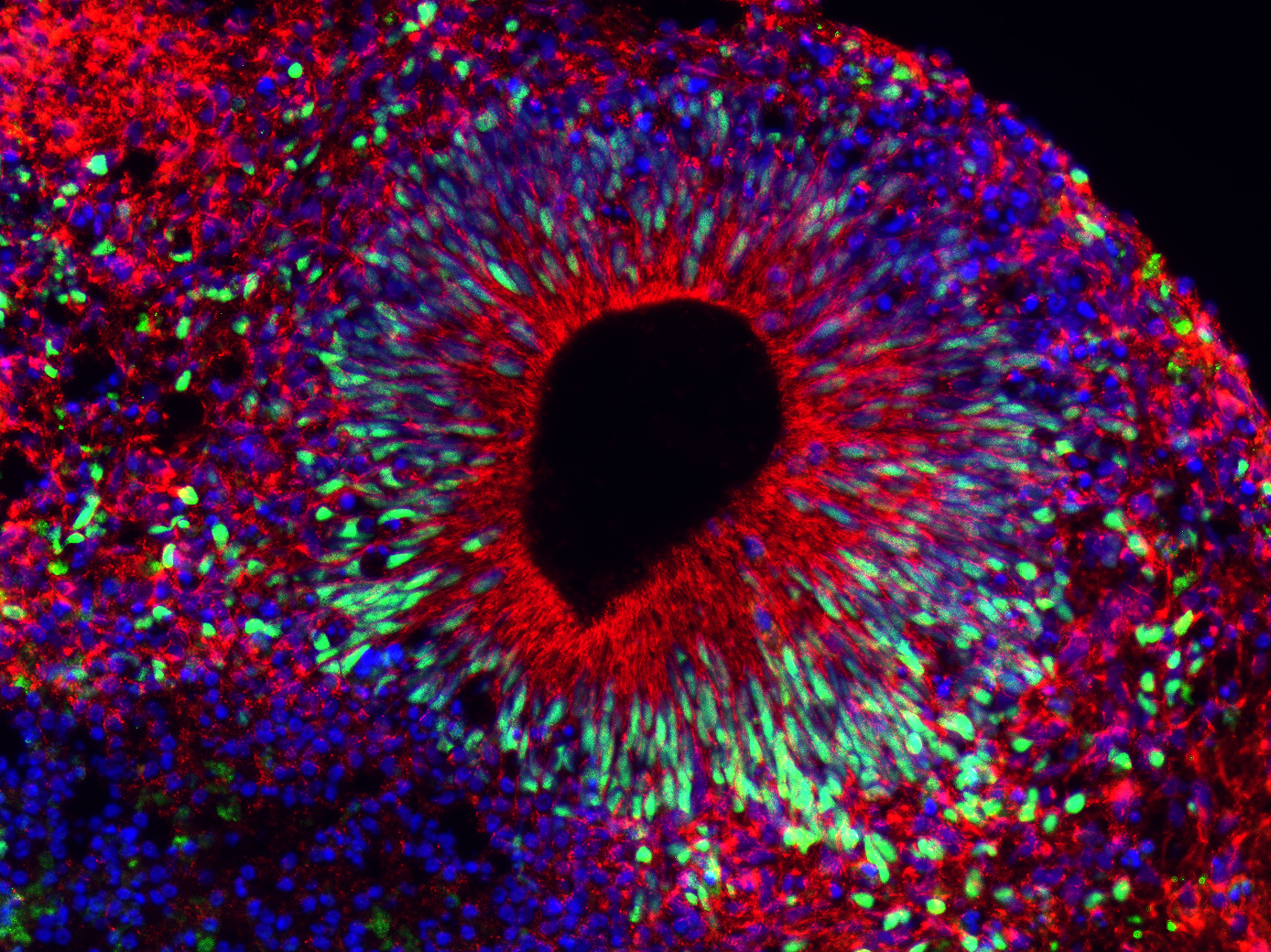
The researchers found that patient-derived brain organoids exhibited reduced growth, deficits in the proliferation of neural progenitor cells and consequential premature neuronal differentiation compared with brain organoids derived from the patient’s unaffected parent, which served as a control. Furthermore, using CRISPR-Cas9-mediated gene editing, the team corrected the genetic variant, which restored normal organoid growth and corrected the neural progenitor cells proliferative deficits.
“We were excited about these results because the patient-derived brain organoids recapitulated the patient’s microcephalic phenotype,” says Dr. Hester. “These findings provide strong evidence that the patient’s genetic variant in AUTS2, which was a single base pair change, contributed to the phenotype observed in the organoids. We then wanted to determine what was happening at the molecular level in these organoids.”
To do so, they analyzed gene expression of individual brain cell types in organoids using single-cell RNA sequencing. The analysis revealed significantly reduced expression of genes in patient brain organoids involved in cell division and the WNT-β-Catenin signaling pathway, a cellular signal transduction pathway that controls neural progenitor cell division and cortical neurogenesis.
“Ultimately, we are performing these studies is to help the patient, but for us to help them, we need to first understand the underlying pathophysiology. The long-term goal and hope is then to translate these research findings into a viable therapy or treatment option for the patient,” adds Dr. Hester.
This study not only uncovered a novel role for AUTS2 in neural progenitor cells during human brain development but also highlights the value of brain organoids to investigate molecular mechanisms in other neurodevelopmental disorders. However, the researchers emphasized that more research is necessary to understand the long-term impact of AUTS2 deficiency during neural differentiation and maturation and how this might be associated with intellectual disability in patients.
“This work wouldn’t have been possible without a team science approach, and here at Nationwide Children’s, we were able to start this research first from the referring physician who observed the patient in the clinic to the genomics team in the Institute for Genomic Medicine that identified the AUTS2 variant in the patient’s genome to my lab, where we are able to model the patient’s variant in organoids and bring some insight into the mechanism underlying AUTS2 syndrome,” shares Dr. Hester. “Having that ability and level of collaboration at a single institution is very rare. That’s one of the reasons why this study was unique and innovative, and successful. It also serves as a case study paradigm for future investigations.”
Dr. Hester and colleagues are now poised to use the brain organoid model as a system to screen potential therapies, including genome editing and gene therapy strategies, for patients with neurodevelopmental disorders.
“Using brain organoids gives us unique molecular insights into human-specific aspects of brain development that can’t be analyzed using animal models,” says Dr. Hester. “For example, mouse models of AUTS2 deficiency do not develop microcephaly. It is important to emphasize that organoids don’t replace animal models and both model systems have advantages and disadvantages that complement each other, although organoids fill the gap in understanding what is occurring in human disease.”
Reference
Fair SR, Schwind W, Julian D, Biel A, Guo G, Rutherford R, Ramadesikan S, Westfall J, Miller KE, Kararoudi MN, Hickey SE, Mosher TM, McBride KL, Neinast R, Fitch J, Lee D, White P, Wilson RK, Bedrosian TA, Koboldt DC, Hester ME. Cerebral organoids containing an AUTS2 missense variant model microcephaly. Brain. 2022 Jul 8:awac244. doi: 10.1093/brain/awac244. Epub ahead of print. PMID: 35802027.
Image credit: Nationwide Children’s (courtesy of Hester Lab)
About the author
Lauren Dembeck, PhD, is a freelance science and medical writer based in New York City. She completed her BS in biology and BA in foreign languages at West Virginia University. Dr. Dembeck studied the genetic basis of natural variation in complex traits for her doctorate in genetics at North Carolina State University. She then conducted postdoctoral research on the formation and regulation of neuronal circuits at the Okinawa Institute of Science and Technology in Japan.
- Lauren Dembeckhttps://pediatricsnationwide.org/author/lauren-dembeck/
- Lauren Dembeckhttps://pediatricsnationwide.org/author/lauren-dembeck/
- Lauren Dembeckhttps://pediatricsnationwide.org/author/lauren-dembeck/
- Lauren Dembeckhttps://pediatricsnationwide.org/author/lauren-dembeck/January 29, 2019







