Cerebral Organoids Provide Insight into Human Brain Development and Neurological Disease
Cerebral Organoids Provide Insight into Human Brain Development and Neurological Disease https://pediatricsnationwide.org/wp-content/uploads/2020/11/organoid-section-HEADER-1024x575.jpg 1024 575 JoAnna Pendergrass, DVM JoAnna Pendergrass, DVM https://pediatricsnationwide.org/wp-content/uploads/2021/03/pendergrass_01.jpg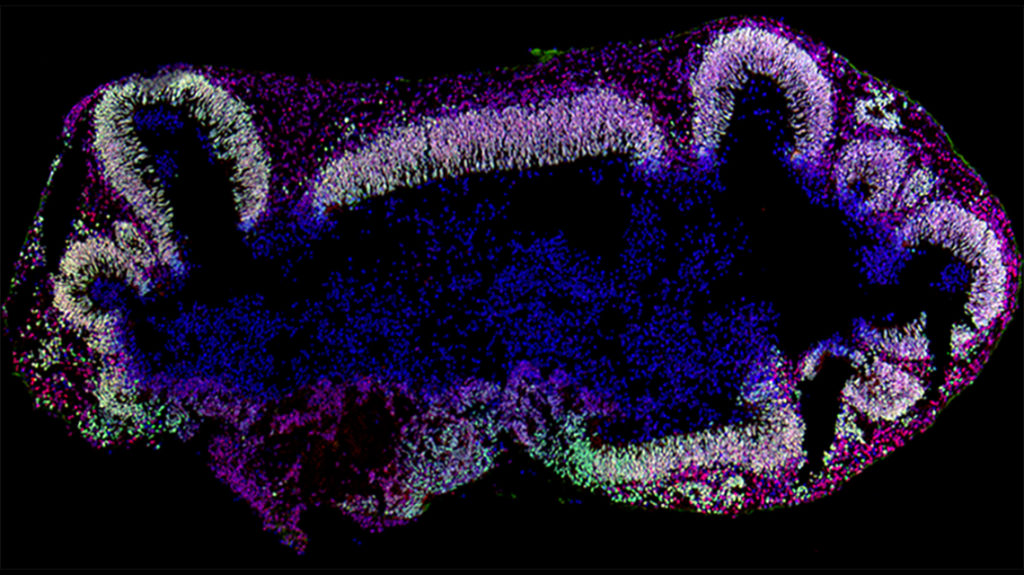
Correlations between the neuroelectrical maturation and cellular development of cerebral organoids highlight the organoids’ translational and therapeutical potential in early neurodevelopmental disorders.
Cerebral organoids are three-dimensional miniature organs that resemble the human brain. Derived from human pluripotent stem cells, cerebral organoids have emerged to advance stem cell research, improve three-dimensional tissue culture techniques and enhance understanding of human fetal brain development and neurological disease.
Rodent models, although mechanistically useful, cannot fully capture the complex morphology, cellular diversity and neural connectivity of the human brain.
“Cerebral organoids represent an innovative model in neuroscience research of the human brain,” says Mark Hester, PhD, a principal investigator in the Steve and Cindy Rasmussen Institute for Genomic Medicine at Nationwide Children’s Hospital.
In the first study of its kind, Dr. Hester and his research team reported correlations between neuroelectrical activities and the molecular and cellular development of cerebral organoids. The study is published in Stem Cell Reports.
“Dr. Hester’s work on cerebral organoids is absolutely critical to clinician-scientists who support the neurodevelopmental progression and treatment of hospitalized infants. His work provides the building blocks we need to test new early treatments for very real clinical problems, from preterm brain injury to prenatal harmful exposures,” says Nathalie Maitre, MD, PhD, neonatologist, principal investigator in the Center for Perinatal Research, director of NICU Follow-up Programs at Nationwide Children’s and co-author of the study.

The study’s findings also highlight the translational and therapeutic potential of cerebral organoids. Understanding the correlation between the neuroelectrical and molecular development in cerebral organoids brings additional value to their ability to model complex neurological disorders. Such insights could lead to targeted therapies that would restore proper electrophysiological function or replace a defective gene, says Dr. Hester.
He also notes that the organoids are an excellent model for understanding molecular mechanisms underlying early neurodevelopmental disorders, such as autism spectrum disorder and epileptic-encephalopathies. In addition, since cerebral organoids can be generated from patients, they can be used to characterize newly identified genetic variants and develop personalized treatments.
Cerebral organoid research could also elucidate the influence of environmental factors, such as viral pathogens, on neuroplasticity in neonatal brain development.
For the study, the organoids were cultured for five months and evaluated at monthly intervals.
The researchers first measured the organoids’ spontaneous neuroelectrical activity using a multi-electrode array (MEA) platform.
“This platform can provide critical insights into the underlying molecular mechanisms of neural connectivity and plasticity,” says Dr. Hester.
Neuroelectrical activity first appeared at approximately day 30, maturing from low-amplitude spiking activity to synchronized burst firings.
Next, the researchers studied the cerebral organoids’ complex developmental changes with single-cell RNA sequencing (scRNA-seq) and immunohistochemistry (IHC).
scRNA-seq identified eight major cell populations, including excitatory and inhibitory neurons. A surge in astrocytes was observed after 2-3 months of culture. Astrocytes influence neuronal connectivity and electrophysiology, Dr. Hester explains.
A key IHC finding was an increase in GABA expression that mirrored a surge in the GABAergic neuron population after several months of culture. The researchers also observed the late emergence of the neurotrophin (NTR)/TRK signaling pathway, which could influence neuroelectrical activity in cerebral organoids.
Currently, Dr. Hester is collaborating with Dr. Maitre and Kristen Benninger, MD, neonatologist at Nationwide Children’s, to investigate how opioid exposure affects human brain development at the molecular and cellular levels. The cerebral organoid model, he explains, will allow exploration into opioids’ effects on brain development timing and opioid-susceptible cell types.
Despite their significant potential, cerebral organoids do have limitations. For example, they do not contain the variety of brain cell types that would allow them to demonstrate complex signaling events. Also, cerebral organoid cultures can be highly variable. However, recent innovations in organoid protocols have begun to address these challenges.
Cerebral organoids represent an advancement in studying human brain development and should be used to complement, rather than replace, existing models, says Dr. Hester.
References
Fair SR, Julian D, Hartlaub AM, Pusuluri ST, Malik G, Summerfied TL, Zhao G, Hester AB, Ackerman IV WE, Hollingsworth EW, Ali M, McElroy CA, Buhimschi IA, Imitola J, Maitre NL, Bedrosian TA, Hester ME. Electrophysiological maturation of cerebral organoids correlates with dynamic morphological and cellular development. Stem Cell Reports. Sep 2020.
Image credits: Nationwide Children’s, Hester Lab
About the author
JoAnna Pendergrass, DVM, is a veterinarian and freelance medical writer in Atlanta, GA. She received her veterinary degree from the Virginia-Maryland College of Veterinary Medicine and completed a 2-year postdoctoral research fellowship at Emory University’s Yerkes Primate Research Center before beginning her career as a medical writer.
As a freelance medical writer, Dr. Pendergrass focuses on pet owner education and health journalism. She is a member of the American Medical Writers Association and has served as secretary and president of AMWA’s Southeast chapter.
In her spare time, Dr. Pendergrass enjoys baking, running, and playing the viola in a local community orchestra.
- JoAnna Pendergrass, DVMhttps://pediatricsnationwide.org/author/joanna-pendergrass-dvm/
- JoAnna Pendergrass, DVMhttps://pediatricsnationwide.org/author/joanna-pendergrass-dvm/
- JoAnna Pendergrass, DVMhttps://pediatricsnationwide.org/author/joanna-pendergrass-dvm/
- JoAnna Pendergrass, DVMhttps://pediatricsnationwide.org/author/joanna-pendergrass-dvm/