Decoding Pediatric Asthma: From Cells to Care
Decoding Pediatric Asthma: From Cells to Care https://pediatricsnationwide.org/wp-content/uploads/2026/04/W1909850-Rsrch_PedsNW-SpringSummer2026_Pg-24-header-1024x448.jpg 1024 448 Madison Storm Madison Storm https://pediatricsnationwide.org/wp-content/uploads/2023/09/092023BT159.png
How clinician-scientist teams are mapping the molecular drivers of wheezing and asthma to guide smarter, faster and more personalized treatment.
Asthma is one of the most common reasons children visit the Emergency Department (ED) at Nationwide Children’s Hospital. While frontline clinicians work quickly to stabilize breathing, researchers across the hospital are probing deeper: What’s happening inside the airway at the cellular level? Why do some children respond to standard treatment while others don’t? How can this knowledge lead to better care?
From point-of-care sample collection during ED visits to advanced cellular modeling and novel preclinical testing, teams at Nationwide Children’s are collaborating to uncover the mechanisms behind asthma severity and treatment resistance.
From Emergency Care to Discovery
For Adjoa Andoh, MD, emergency medicine physician at Nationwide Children’s, the questions begin the moment a child arrives with respiratory distress.
“When a child comes in with severe asthma, it’s all hands on deck,” says Dr. Andoh. “The first thing we do is assess the severity of their asthma and start standard treatments, such as bronchodilators and steroids, but families often ask, ‘Why isn’t my child responding [to treatment]?’ and right now, we don’t have a clear answer. We just know some kids respond and some don’t. That’s where the research comes in.”
Practitioners rely on physical exam findings and real-time responses to medications, but this model has limits.
“We have no way to clinically predict who will improve and who won’t. That’s frustrating for families and for us,” says Dr. Andoh.
These gaps sparked a collaboration with two research teams: Katherine Bline, MD, critical care physician and principal investigator in the Center for Microbe and Immunity Research, and Rodney Britt, PhD, ATSF, principal investigator in the Center for Perinatal Research.
Together, they launched Pathophysiology of Asthma-Induced Critical Illness (PACI), which is an innovative project collecting and analyzing blood samples from children across the spectrum of asthma severity. These samples may reveal biomarkers that predict treatment response, paving the way for personalized care.
“We want to collect samples early, before treatments alter their immune profile,” explains Dr. Andoh. “There have been very few studies that look at what’s happening immunologically during an acute asthma exacerbation; it’s almost exploratory in nature.”
Inside the ICU: When Asthma Turns Critical
While many patients improve with standard ED care, others require intensive support. In the Pediatric Intensive Care Unit (ICU), Dr. Bline sees the most severe end of the asthma spectrum.
“For kids with asthma, being hospitalized is a big deal. Some improve with additional treatment, but others persist or become more severe,” says Dr. Bline. “There’s an enormous spectrum of illness severity.”
Dr. Bline’s research focuses on myeloid-derived suppressor cells (MDSCs) — immune regulators that may play a dual role depending on context.
“In infection, MDSCs can be harmful because they suppress T cells,” she explains. “But in asthma, where there’s a lot of inflammation, MDSCs might help bring the lungs back toward immune homeostasis. Some patients may have MDSCs that simply aren’t doing what they need to do, especially in the context of steroids.”
Dr. Bline’s team investigates how these immune cells behave across asthma severities and whether their properties might predict steroid resistance.
“The ultimate goal is precision medicine,” says Dr. Bline. “If we can define distinct immunologic phenotypes, we can tailor therapies instead of relying on a one-size-fits-all approach.”
Why Some Don’t Respond to Steroids
From a mechanistic standpoint, one of the common questions in asthma care is why corticosteroids, the foundation of acute treatment, work from some but not for others.
For Dr. Britt, this question is central. His lab studies inflammatory regulator in airway smooth muscle, the tissue that controls airway tone (how well the airways open and close) and contributes to obstruction when it contracts and thickens.
Using human primary airway smooth muscle cells, immune cells and allergen mouse models, his team examines how different inflammatory pathways (Type 1, Type 2 and Type 17) change airway smooth muscle behavior and shape asthma severity.
“We’ve treated airway smooth muscle cells with cytokines from each pathway alone and in combination, and then in the presence of steroids,” explains Dr. Britt. “In complex inflammatory environments, airway smooth muscle hyperresponsiveness and thickening persist despite steroid treatment. Meanwhile, the eosinophil is highly sensitive to steroids, but the T cell populations driving more severe disease behave differently. Steroids don’t impact neutrophils the way they do eosinophils. That’s a key part of why steroid-resistant asthma exists.”
Dr. Britt’s team is also exploring combination therapies.
His long-term vision includes defining the molecular intersections among pathways to uncover targeted treatments.
That complexity becomes even more pronounced during virus-triggered attacks, one of the most common drivers seen in the ED and ICU. Viral infections can tilt immune pathways toward mixed, steroid-insensitive patterns and may even program long-term risk in early life.
Viruses and the Asthma Connection
Respiratory viruses are a major trigger for asthma attacks and may even shape long-term risk. Mitchell Grayson, MD, chief of the Division of Allergy and Immunology and principal investigator in the Center for Clinical and Translational Research, is studying how early life respiratory viral infections influence wheezing and can drive the development of asthma.
“Infants hospitalized with severe RSV have an increased risk of recurrent wheeze and possibly even developing asthma,” says Dr. Grayson. “The question is: why would a viral infection drive allergic disease?”
Dr. Grayson’s lab has shown that mice with viral infections produce IgE antibodies — the same antibodies involved in allergic responses — against respiratory viruses.
“In our models, we’ve found that mice make IgE (the allergic antibody) against the virus, and that’s required for them to develop asthma-like symptoms.”
His team uses Sendai virus (murine parainfluenza type 1), respiratory syncytial (RSV) and human bronchial epithelial cell cultures to model human disease mechanisms.
“Almost all asthma attacks are virally induced,” emphasizes Dr. Grayson. “Understanding virus-specific immune responses could help us interrupt the path from infection to chronic asthma, as well as preventing subsequent exacerbations in those with asthma.”
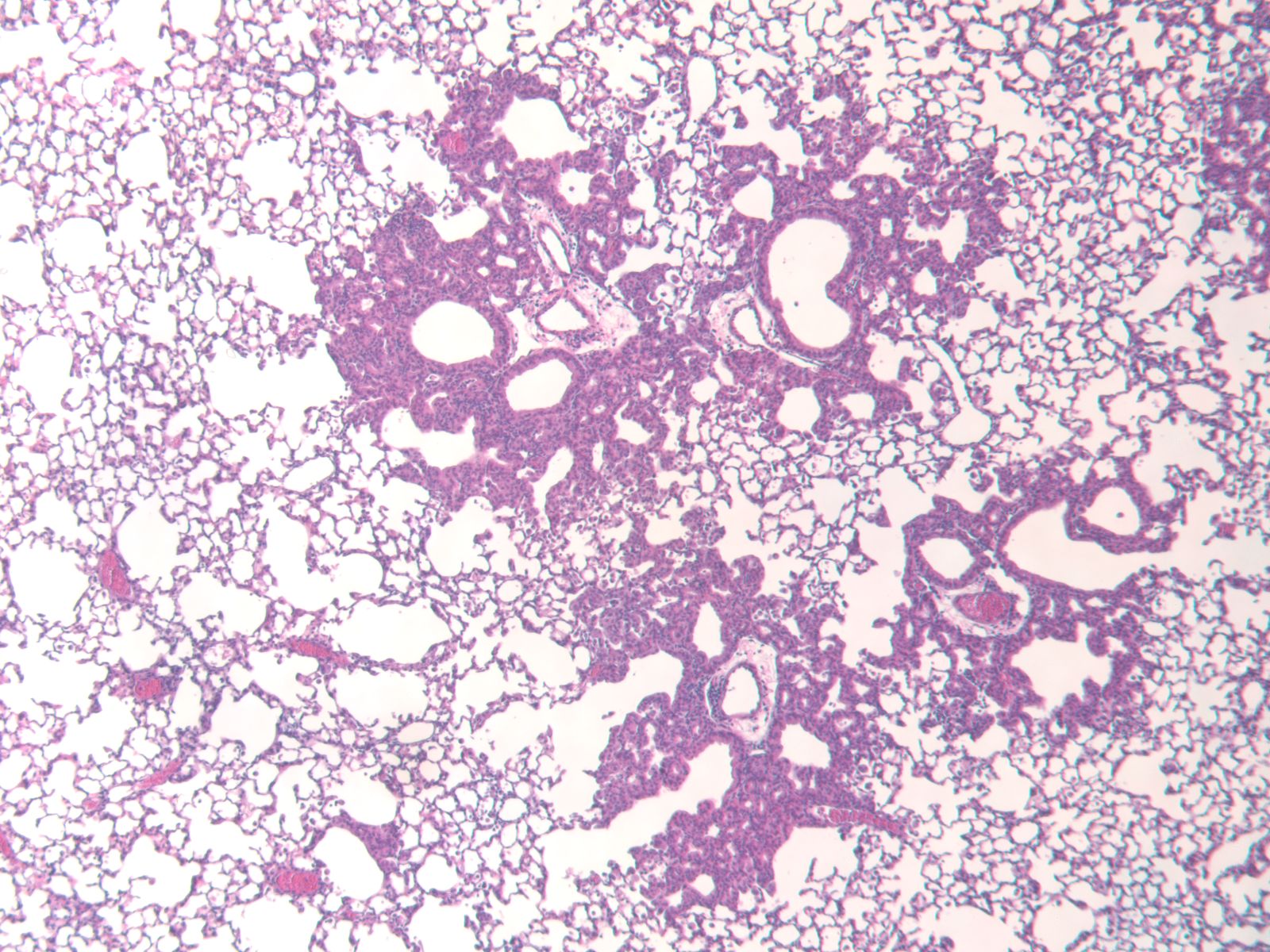
Too Much Mucus: Untangling Airway Epithelium Biology
Another critical player in asthma is mucus, specifically, too much of it. Excess mucus can block small airways, making breathing difficult. In the Center for Perinatal Research, Susan Reynolds, PhD, principal investigator, studies the airway epithelium — the cells lining the respiratory tract — to understand why this happens.
“Asthma is one of several diseases that are characterized as muco-secretory lung disease,” says Dr. Reynolds. “The airway epithelium produces too much mucus, and in the small airways, that mucus can plug the tube completely. That prevents air from getting in and carbon dioxide from getting out. Physiologically, the lung changes its function in response [to the carbon dioxide].”
Her lab focuses on basal cells, the originator of goblet cells that produce mucus.
Recent work has revealed a surprising twist in the Notch signaling pathway.
“We’ve discovered that Notch ligands are not just activating the receptor — they’re undergoing processing within the ligand-expressing cell,” says Dr. Reynolds. “Our data suggests that ligand processing regulates whether that cell will change its fate. It’s a novel sub-mechanism, and we’re excited about it.”
Dr. Reynolds’ work could inform therapies not only for asthma, but also for conditions such as cystic fibrosis and chronic obstructive pulmonary disease.
Modeling Human Airways More Accurately
Most preclinical models lack the mucus producing glands needed to effectively study how they influence the development of asthma — and how they respond to potential treatments. In response to this challenge, Thomas Lynch, PhD, principal investigator in the Center for Perinatal Research, has developed an allergic airway inflammation model in ferrets that is now available to colleagues across the research institute.
“Ferrets have airway glands similar to humans and out initial experiments have been really promising,” says Dr. Lynch. “Structural changes like goblet cell hyperplasia and gland hypertrophy appear quickly. This model helps us test therapies that target mucus production in a system that mirrors human anatomy.”
Beyond mirroring human airway structure, the ferret model gives teams a shared platform to probe questions raised elsewhere. For example, how early-life respiratory viruses might ‘set the stage’ for later wheeze and asthma, a central focus of Dr. Grayson’s group. By layering controlled viral exposures onto allergic inflammation, investigators can test whether virus-driven immune responses shift mucus-gland growth, airway smooth muscle behavior and steroid responsiveness — before moving to human studies.
This model also complements Dr. Reynolds’ epithelial work by allowing researchers to examine how Notch-guided basal-to-goblet-cell decisions play out in an intact, gland-rich airway and whether candidate therapies that modulate those pathways reduce mucus plugging in vivo.
“This model gives us a platform to study those pathways and test therapies before moving into human trials,” says Dr. Lynch.
Just as importantly, the readouts from this model (structural changes, immune signatures and treatment responses) feed back to the bedside. As biomarkers and candidate steroid-sparing combinations emerge, Drs. Andoh and Bline can integrate them into ED and ICU studies such as PACI to stratify children earlier, avoid unnecessary side effect and escalate the right care faster.
This article appeared in the 2026 Spring/Summer print issue. Download the issue here.
References:
- Ford ML, Reza MI, Ruwanpathirana A, Sathish V, Britt RD. Integrative roles of pro-inflammatory cytokines on airway smooth muscle structure and function in asthma. Immunological Reviews. 2025;330(1):e70007.
- Reynolds SD, Hill CL, Alsudayri A, Lallier SW, Wijeratne S, Tan ZH, Chiang T, Cormet-Boyaka E. Assemblies of JAG1 and JAG2 determine tracheobronchial cell fate in mucosecretory lung disease. JCI Insight. 2022 Aug 8;7(15):e157380.
Image credits: Nationwide Children’s
About the author
Madison Storm is the Senior Strategist of Research Communications at Nationwide Children's Hospital. She earned her bachelor's in multimedia journalism from Virginia Tech in 2021 and went on to achieve her master's in health communication from Johns Hopkins University in 2023. Her passion for transforming the complex to clear is supported by various experiences writing for consumer audiences.
- Madison Stormhttps://pediatricsnationwide.org/author/madison-storm/
- Madison Stormhttps://pediatricsnationwide.org/author/madison-storm/February 27, 2024
- Madison Stormhttps://pediatricsnationwide.org/author/madison-storm/
- Madison Stormhttps://pediatricsnationwide.org/author/madison-storm/
- Posted In:
- Features
- In Sight
- Research
- Uncategorized














