Choosing a Continuous Glucose Monitor After TPIAT: When Research Directly Impacts Clinical Practice
Choosing a Continuous Glucose Monitor After TPIAT: When Research Directly Impacts Clinical Practice https://pediatricsnationwide.org/wp-content/uploads/2020/10/AdobeStock_313527520_for-Web-1024x683.jpg 1024 683 Pam Georgiana Pam Georgiana https://pediatricsnationwide.org/wp-content/uploads/2023/07/May-2023.jpg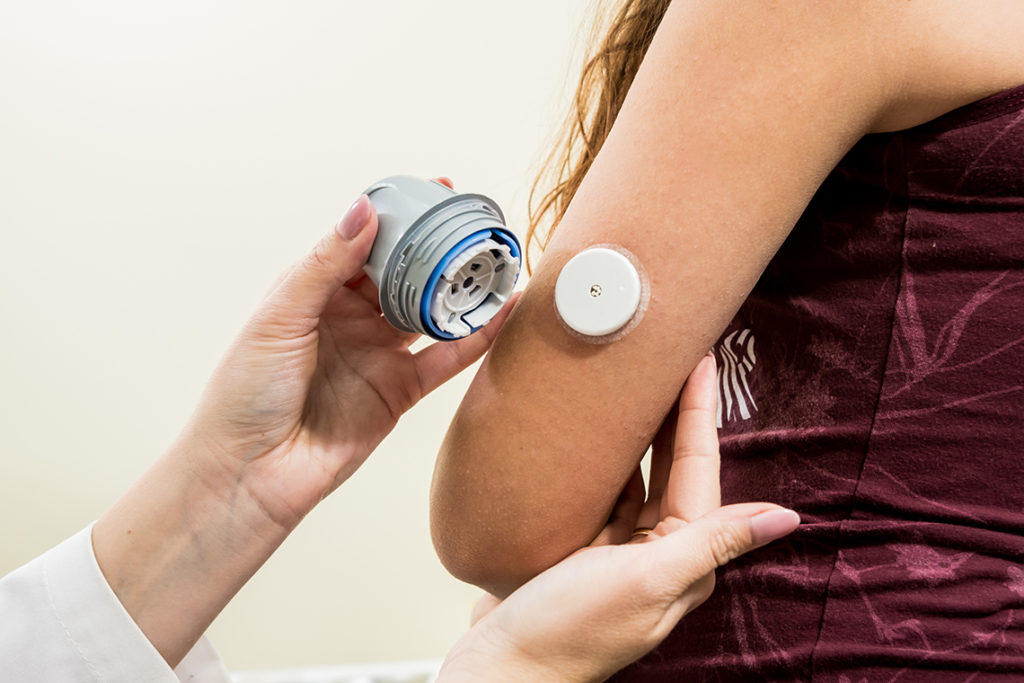
Nationwide Children’s study highlights hydroxyurea interference with Dexcom, but not with Libre, and the clinical implications for glycemic management.
Total pancreatectomy with islet autotransplantation (TPIAT) is the definitive surgical treatment for acute recurrent and chronic pancreatitis. Nationwide Children’s Hospital is one of the very few pediatric medical centers in the nation to offer this complex procedure.
By removing the pancreas, TPIAT relieves pain caused by pancreatitis. However, pancreatectomy also eliminates endogenous insulin production, resulting in immediate diabetes. Islet autotransplantation mitigates this risk by purifying islets from the resected pancreas and infusing them into the portal vein to the liver, where they may engraft and resume insulin secretion. To support islet survival, patients require strict glycemic management after TPIAT, using wearable insulin pumps and continuous glucose monitors (CGMs).
Hydroxyurea is routinely prescribed after TPIAT to reduce reactive thrombocytosis, an abnormally high platelet count that increases the risk of clotting in the portal vein. However, prior studies have shown that this medication interferes with the enzymatic chemistry used in one particular CGM (Dexcom), causing falsely high readings. While the FreeStyle Libre CGM was reported not to have similar interference, its accuracy had never been evaluated in children after TPIAT.
To evaluate the effectiveness of these two popular CGMs in this population, Jennifer M. Ladd, MD, MSc, a physician in the Division of Endocrinology at Nationwide Children’s, and colleagues from the TPIAT and Diabetes/Insulin Inpatient Service Consult (DIISC) teams conducted a prospective observational study comparing Dexcom and Libre performance in pediatric TPIAT patients receiving hydroxyurea. Diabetes Technology & Therapeutics recently published the results.
“Strict glucose management in the immediate postoperative period is essential to protect islet engraftment. If our monitoring technology is unreliable under these conditions, it directly impacts patient care,” Dr. Ladd explains.
For the study, seven pediatric participants (ages 8 to 20) recovering from TPIAT wore both Dexcom and Libre devices, starting approximately one week post-surgery. Simultaneous point-of-care (fingerstick) testing was also performed for reference. The team reviewed 560 paired measurements. They calculated absolute and relative differences, as well as the mean absolute relative difference (MARD). They used Clarke error grid analysis to evaluate clinical accuracy.
In the nine hours following hydroxyurea administration, Dexcom values were significantly higher than those from fingerstick measurements, while the Libre device tracked closely. Only 77.9% of Dexcom readings fell within a clinically acceptable range, compared to 98.3% for Libre.
Outside the immediate hydroxyurea window, both devices performed more reliably. MARD for Dexcom was 19.8% compared to 14.7% for Libre, and both systems achieved an acceptable clinical agreement with fingerstick values.
“The key finding is that hydroxyurea consistently interferes with Dexcom accuracy, causing false hyperglycemia, but does not interfere with Libre,” Dr. Ladd explains. “Use of Dexcom in this setting could lead to inappropriate insulin dosing causing hypoglycemia, excess fingersticks, and greater patient burden.” In contrast, Libre maintained accuracy under identical conditions.
The study is among the first to provide direct, clinically applicable evidence for device selection in the TPIAT population.
“We cannot endorse one commercial device over another, but these data allow our families and clinicians to weigh risks and benefits when choosing glucose monitoring tools,” Dr. Ladd emphasizes.
The findings confirm that hydroxyurea significantly impairs Dexcom accuracy, but does not affect Libre. For pediatric patients recovering from TPIAT who require hydroxyurea, Libre may better support glucose management and ultimately islet engraftment.
“Studies like this remind us how critical it is to evaluate medical technologies in the specific populations we serve,” says Dr. Ladd. “At Nationwide Children’s, we are uniquely positioned to both provide advanced procedures like TPIAT and to generate the research needed to ensure the tools we use are safe and effective for these children. That dual commitment to care and discovery drives better outcomes for our patients and for the field.”
Reference:
Ladd JM, Gandhi K, Friesner-Gephart K, Hoffman RP, Okafor MJ, Heinzman C, Gariepy CE, Rasmussen SK, Nathan JD, Freeman AJ. Accuracy of Two Continuous Glucose Monitors Differs after Hydroxyurea in Pediatric Patients Undergoing Total Pancreatectomy with Islet Autotransplantation. Diabetes Technol Ther. 2025 Aug;27(8):651-658.
About the author
Pam Georgiana is a brand marketing professional and writer located in Bexley, Ohio. She believes that words bind us together as humans and that the best stories remind us of our humanity. She specialized in telling engaging stories for healthcare, B2B services, and nonprofits using classic storytelling techniques. Pam has earned an MBA in Marketing from Capital University in Columbus, Ohio.
- Pam Georgianahttps://pediatricsnationwide.org/author/pam-georgiana/
- Pam Georgianahttps://pediatricsnationwide.org/author/pam-georgiana/
- Pam Georgianahttps://pediatricsnationwide.org/author/pam-georgiana/
- Pam Georgianahttps://pediatricsnationwide.org/author/pam-georgiana/
- Posted In:
- Uncategorized