Using Hormone Therapy First Drives Earlier Remission From Infantile Epileptic Spasms Syndrome
Using Hormone Therapy First Drives Earlier Remission From Infantile Epileptic Spasms Syndrome https://pediatricsnationwide.org/wp-content/uploads/2020/08/mother-and-infant-header-1024x575.gif 1024 575 Natalie Wilson Natalie Wilson https://pediatricsnationwide.org/wp-content/uploads/2021/06/Natalieheadshot3-2.png
Recent research conducted at Nationwide Children’s Hospital demonstrated standardizing hormone therapy as the first treatment for infantile spasms improved rates of early remission.
Infantile epileptic spasms syndrome (IESS) is an uncommon epilepsy syndrome characterized by seizures called infantile spasms that begin between 1 and 24 months of age (with a peak age of onset at 6 months) and an electroencephalogram (EEG) that is typically consistent with an epileptic encephalopathy. IESS is associated with numerous genetic conditions, brain malformation, injuries and a variety of brain diseases. In the United States, there are 2,000 to 2,500 new cases of IESS each year.
Children with IESS often have poor development and medically refractory epilepsy; however, early and effective treatment can improve these outcomes for many children. The best outcomes are possible when that treatment enables complete remission from infantile spasms and resolution of the epileptic encephalopathy on EEG.
The American Academy of Neurology and Child Neurology Society have strongly recommended that one of three standard medications be used as the first treatment for IESS: adrenocorticotropic hormone (ACTH), vigabatrin or high-dose oral prednisolone. Because there is no recommendation that one of the medications be selected over the others, there is significant variability in clinical practice around the world.
Available evidence suggests, however, that selecting either ACTH or prednisolone, hormone therapies, as the first treatment over vigabatrin is associated with higher rates of early remission. And just over half of children with IESS enter remission within 3 months of the start of initial treatment.
Using quality improvement (QI) methodology that standardized hormone therapy as the first treatment, researchers at Nationwide Children’s Hospital increased the rate of 3-month remission of children with IESS from about 54% to nearly 76%.
The QI project, recently published in Neurology, included several interventions to improve clinical practice, but John Mytinger, MD, director of the Infantile Spasms Program and lead author of the study, identified this treatment prioritization as the major driver of improvement.
“In our baseline cohort, vigabatrin was the most frequently used first treatment. We made a systematic, evidence-based change to use hormone therapy as the first treatment for all eligible children in our intervention cohort, and outcomes significantly improved,” says Dr. Mytinger, who is also an assistant professor of Pediatric Neurology at The Ohio State University College of Medicine. “We improved local outcomes simply by optimizing the use of currently available treatments. Similar QI interventions at other centers may improve their local remission rates as well.”
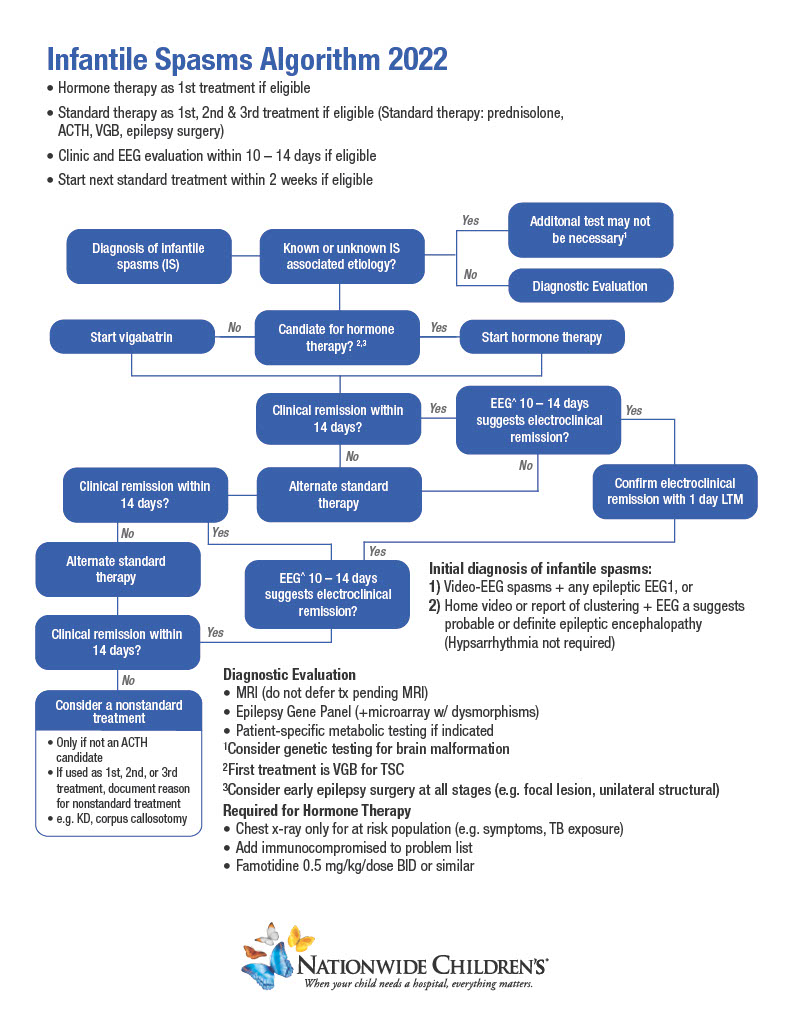
Nationwide Children's Infantile Spasms Protocol Infographic
Learn more about the QI interventions that improved rates of early remission.
Download a high resolution copy. ›Additionally, expert consensus is if the first therapy selected does not stop the infantile spasms on or before treatment day 14, a second treatment should be initiated. As part of this QI project, Dr. Mytinger and his team also standardized earlier follow-up after initiating treatment to facilitate this initiation of a second treatment when necessary. This practice was further supported by a multicenter study published concurrently in Neurology by Dr. Mytinger and other infantile spasms experts that showed remission for most children with IESS is determined after about one week of treatment. Of 395 infants in this study, 138 (about 80% of responders) experienced remission within a week. Only 33 children (about 20% of responders) did not respond within the first week of treatment and experienced remission during the second week.
“We know we need to see these children soon after treatment initiation, ideally within that second week, to balance giving the first medication time to work with quickly enabling the initiation of a second treatment if needed,” says Dr. Mytinger. “For children with IESS, delaying successful treatment may worsen developmental outcomes. Both starting with the right treatment and facilitating early sequential treatment may improve outcomes.”
Dr. Mytinger says he and his team plan to further examine the early initiation of a second treatment for non-responders and hope to conduct a study that evaluates outcomes, such as the rate of uncontrolled epilepsy, at two years of age.
“Because inadequately treated IESS often results in later medication-resistant epilepsy, standardizing hormone therapy at other centers, including those in other countries, has the potential to significantly improve the lives of many young children with epilepsy,” says Dr. Mytinger.
References:
- Mytinger JR, Parker W, Rust SW, Ahrens SM, Albert DV, Beatty CW, Chrisman J, Clark DJ, Debs A, Denney D, Karn M, Herbst J, Ostendorf AP, Taylor MC, Twanow J, Vidaurre J, Patel AD. Prioritizing hormone therapy over vigabatrin as the first treatment for infantile spasms: A quality improvement initiative. Neurology. 2022 Aug 29. Epub ahead of print.
- Yuskaitis CJ, Mytinger JR, Baumer FM, Zhang B, Liu S, Samanta D, Hussain SA, Yozawitz EG, Keator CG, Joshi C, Singh RK, Bhatia S, Bhalla S, Shellhaas R, Harini C; Pediatric Epilepsy Research Consortium. Association of time to clinical remission with sustained resolution in children with new-onset infantile spasms. Neurology. 2022 Aug 29. Epub ahead of print.
About the author
Natalie is a passionate and enthusiastic writer working to highlight the groundbreaking research of the incredible faculty and staff across Nationwide Children's Hospital and the Abigail Wexner Research Institute. Her work at Nationwide Children's marries her past interests and experiences with her passion for helping children thrive and a long-held scientific curiosity that dates back to competing in the Jefferson Lab Science Bowl in middle school. Natalie holds a bachelor’s degree in sociology from Wake Forest University, as well as minors in women's, gender & sexuality studies and interdisciplinary writing. As an undergraduate student, Natalie studied writing and journalism, engaged with anthropological and sociological research with a focus on race and ethnic relations, served as executive editor for the student newspaper, the Old Gold & Black, and gained marketing experience as an intern for a nonprofit entrepreneurial incubator, Winston Starts, as well as by working for Wake Forest University School of Law Office of Communication and Public Relations and its Innocence and Justice Clinic.
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Posted In:
- Clinical Updates
- In Brief
- Research