The Impact of Genomic Medicine: Carter’s Story
The Impact of Genomic Medicine: Carter’s Story https://pediatricsnationwide.org/wp-content/uploads/2021/09/Carter6_rotate_crop-1024x685.jpg 1024 685 Natalie Wilson Natalie Wilson https://pediatricsnationwide.org/wp-content/uploads/2021/06/Natalieheadshot3-2.png
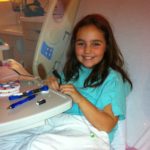
In 2013, at just 1 year old, Carter Daggett was diagnosed with a rare brain tumor. Now, in 2021, at 8 years old, he celebrated three years cancer free — thanks to his clinical teams and cutting-edge research by the team at the Steve and Cindy Rasmussen Institute for Genomic Medicine at Nationwide Children’s Hospital.
Without genomic testing, we would be treating Carter’s tumor based on our best guesses. Knowing the exact mutation we needed to target helped us to quickly arrive at an effective treatment for him. This is precision medicine at its best.
— Nicholas Yeager, MD

“Cancers result from changes to the genomic DNA in our cells,” says Elaine Mardis, PhD, who leads the Institute for Genomic Medicine along with Richard Wilson, PhD, as co-executive director. “DNA, essentially, encodes the instructions for how cells should work. If those instructions get scrambled or damaged, then cells may grow out of control, resulting in a cancer.”
“Genomic medicine uses DNA sequencing to study the DNA from cancer cells and compare it to DNA from normal cells. This allows us to identify the genetic mutations that are the root molecular cause for a patient’s disease and determine precisely what type of cancer we’re fighting, which in turn may suggest specific therapies designed to shut down that root cause,” adds Dr. Mardis.
“It’s much easier to fight an enemy you know and understand, and that’s what allowed for improved care for Carter,” says oncologist Nicholas Yeager, MD, one of Carter’s many physicians at Nationwide Children’s.
Because the Institute for Genomic Medicine identified the mutation responsible for Carter’s tumors, his doctors were able to prescribe the targeted treatment for that mutation that has helped him to stay in remission. He tolerates his medication without side effects, and he hasn’t experienced any developmental delays or academic or social challenges because of his treatments, tumors or surgeries.
“Without genomic testing, we would be treating Carter’s tumor based on our best guesses. Knowing the exact mutation we needed to target helped us to quickly arrive at an effective treatment for him. This is precision medicine at its best,” says Dr. Yeager, who is also director for Oncology in the Division of Hematology, Oncology and Blood & Marrow Transplant and a member of the comprehensive sarcoma program at Nationwide Children’s Hospital and The Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute (OSUCCC – James).
“We have evidence of the highly beneficial impact genomic medicine can have on patients’ care and quality of life,” says Dr. Mardis.
Long Road to Recovery
Carter was a healthy newborn who met all the expected developmental milestones. But when his family took him to the doctor for his 1-year-old checkup appointment, his pediatrician noted that his head seemed large.
“Carter was born at almost 11 pounds, so he was always kind of a big baby. We hadn’t thought anything of it,” says his mother, Liz.

In an abundance of caution, Carter’s pediatrician referred him for a computerized tomography (CT) scan to have X-ray images taken from different angles around his head.
The CT scan revealed Carter had a brain tumor.
Carter successfully underwent surgery to have his tumor removed. Additional testing showed it was a sarcoma, a rare cancerous tumor that occurs in bone, muscle or connective tissues.
Carter’s family and care team decided to follow up his surgery with proton radiation therapy, which was also successful. His brain tumor was removed, and his cancer was controlled.
But in 2018, just five years later, Carter’s doctors detected a reoccurrence. He had another tumor. The initial treatment plan was another surgery and another five-to-seven rounds of radiation to treat it.
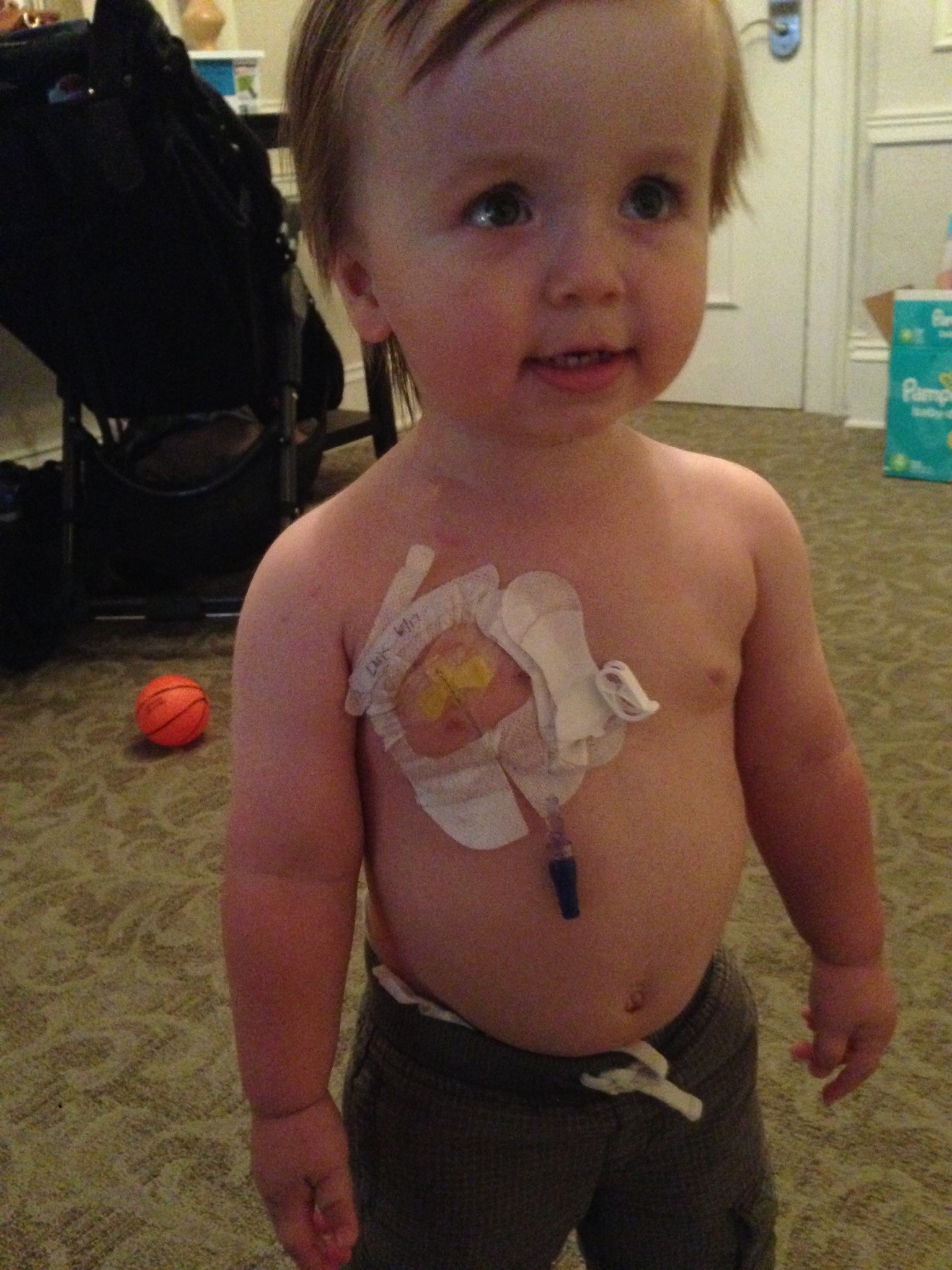
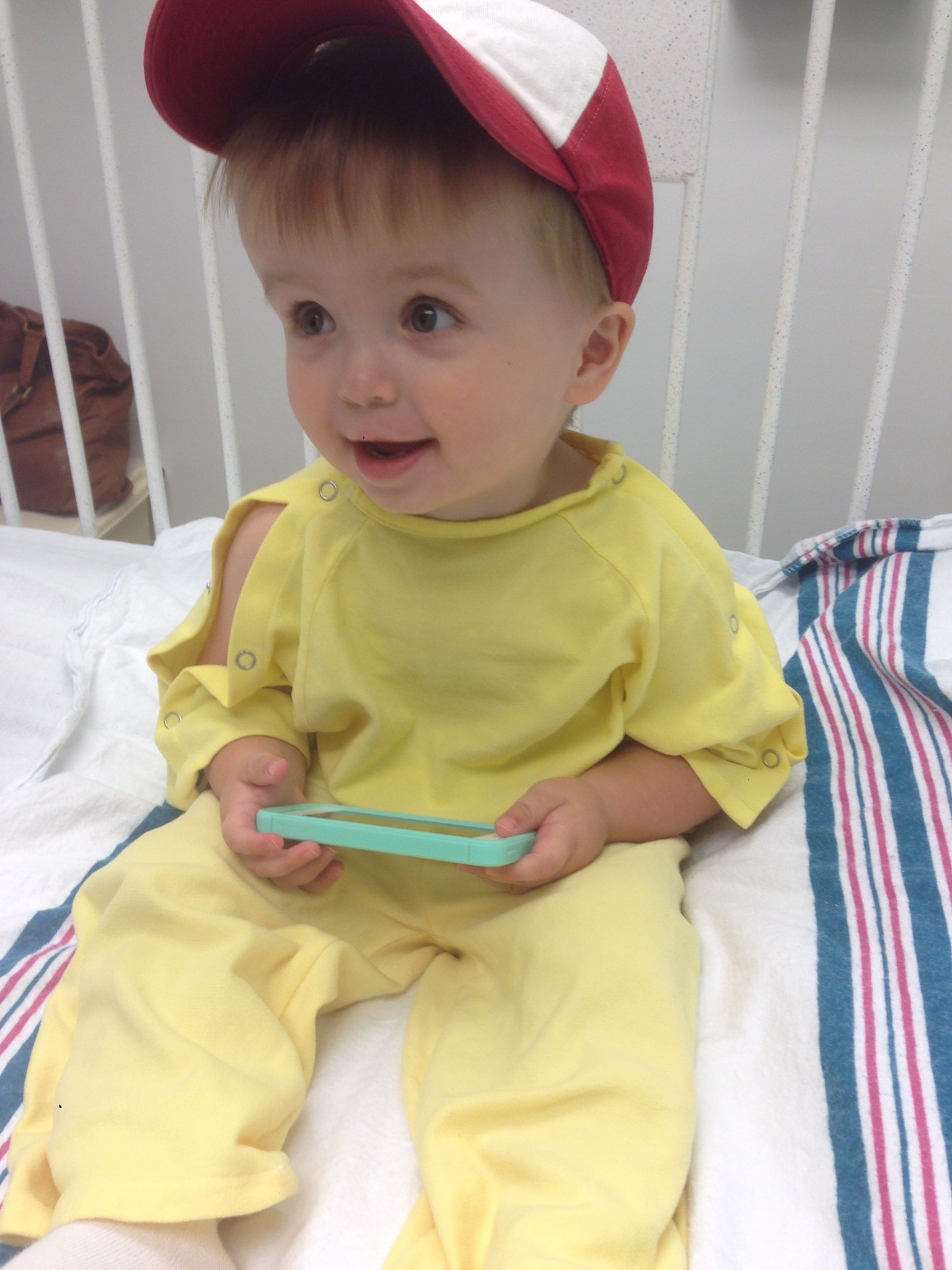
Breaking the Cycle
Carter’s second surgery and treatment were just as successful as his first. So why had his tumor recurred? And how could his doctors try to prevent it from happening again?
Dr. Yeager knew that he and Carter’s family needed more answers. He nominated Carter for a genomic analysis-based research study through the Institute for Genomic Medicine, which had not been available when Carter was first diagnosed. In fact, Carter was only the twenty-first patient nominated for this genomic study.
Genomics provides more certainty in identifying genetic changes, refining diagnoses and suggesting new therapies. It’s also valuable for parents and patients to understand the answer to the question they often want most to know: ‘why?’ Even the basic understanding of why a disease occurred can be critically important.
— Elaine Mardis, PhD

In his nomination, Dr. Yeager noted that Carter’s recurrent sarcoma was “unusual.” The Institute for Genomic Medicine results further classified the sarcoma as a meningeal sarcoma — an especially rare and aggressive tumor that predominantly affects children — and, importantly, identified its cause: a mutation that makes the PDGFRB gene overactive.
“Published research on meningeal sarcomas, their biological behavior and how to manage them clinically is lacking, and they can’t be differentiated from other solid brain tumors using imaging alone. Being able to perform this analysis using tissue from Carter’s tumor to identify his specific mutation was vital,” says Dr. Yeager.
Carter’s clinical team knew the overactivity of the PDGFRB gene could be targeted with a tyrosine kinase inhibitor, an oral chemotherapy drug typically used to treat leukemia patients whose cancers were also associated with the overactivation of this gene.
“Because we know to use this specific class of medications for Carter’s gene mutation, he doesn’t have to be on a lot of different medications. He can be on the one that works,” adds Dr. Yeager.
“It’s rare for a pediatric institution to have the robust genomics program we have here at Nationwide Children’s,” says Dr. Mardis. “Our ability to combine state-of-the-art testing with advanced data analysis is a key strength of our translational studies that can add precision to medical diagnoses.”
“Genomics provides more certainty in identifying genetic changes, refining diagnoses and suggesting new therapies. It’s also valuable for parents and patients to understand the answer to the question they often want most to know: ‘why?’ Even the basic understanding of why a disease occurred can be critically important,” adds Dr. Mardis.
“We’re very lucky to have the team led by Drs. Mardis and Wilson,” says Dr. Yeager. “Integrating laboratory-based research and clinical care is vital to making the right diagnoses and providing the right care. And every day, the Institute for Genomic Medicine is continuing to advance and improve clinicians’ ability to provide that best care for patients like Carter.”


Image credit: Nationwide Children’s (Drs. Yeager and Mardis) and Liz Squires Daggett (Carter)
About the author
Natalie is a passionate and enthusiastic writer working to highlight the groundbreaking research of the incredible faculty and staff across Nationwide Children's Hospital and the Abigail Wexner Research Institute. Her work at Nationwide Children's marries her past interests and experiences with her passion for helping children thrive and a long-held scientific curiosity that dates back to competing in the Jefferson Lab Science Bowl in middle school. Natalie holds a bachelor’s degree in sociology from Wake Forest University, as well as minors in women's, gender & sexuality studies and interdisciplinary writing. As an undergraduate student, Natalie studied writing and journalism, engaged with anthropological and sociological research with a focus on race and ethnic relations, served as executive editor for the student newspaper, the Old Gold & Black, and gained marketing experience as an intern for a nonprofit entrepreneurial incubator, Winston Starts, as well as by working for Wake Forest University School of Law Office of Communication and Public Relations and its Innocence and Justice Clinic.
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Posted In:
- Patient Story