How to Make a Viral Vector
How to Make a Viral Vector https://pediatricsnationwide.org/wp-content/themes/corpus/images/empty/thumbnail.jpg 150 150 Abbie Miller Abbie Miller https://pediatricsnationwide.org/wp-content/uploads/2023/05/051023BT016-Abbie-Crop.jpgThe Research Institute at Nationwide Children’s Hospital is the home to a current Good Manufacturing Practices (cGMP) Clinical Manufacturing Facility (CMF) that operates according to FDA cGMP Guidelines to ensure the safety of manufactured biologic products.
The Space
The CMF is a 9000-sq-ft space, including a 7500-sq-ft clean room suite with ISO Class 5/7/8 spaces and 1500-sq ft quality control lab and research production spaces. The pressurization of the central corridor in the viral vector production suite allows the concurrent production of four distinct viral vector products. This allows the release of vector products for multiple clients in an expedited timeline. Biological drug substances are manufactured according to the FDA Guidance for Industry cGMP for phase I investigational drugs, to ensure product safety, identity, purity and strength.
Production Capacity
The current capacity for a single lot of AAV production is 20 36-layer HYPERstacks, which creates 6 billion cells ready for viral transfection and can yield upwards of 1E+15 vg/mL. The CMF can run three concurrent campaigns (lots) at one time.
The Process: Transfection
Transfection is the process by which the viruses acquire the modified genetic material, in this case, DNA. Transfection occurs in HYPERStack trains – groups of stacks of plastic cases wherein layers of HEK293 cells grow. HEK293 cells are a cell line derived from human embryonic kidney cells that are grown in tissue cultures.
Through transient transfection, engineered plasmid DNA is introduced to the cells along with calcium chloride, sodium phosphate and HEPES, an organic chemical buffering agent. The HEK293 cells then function as small virus-producing factories. Once the process is complete, our team harvests the product and prepares it for purification by clarifying and concentrating the volume using tangential flow filtration.
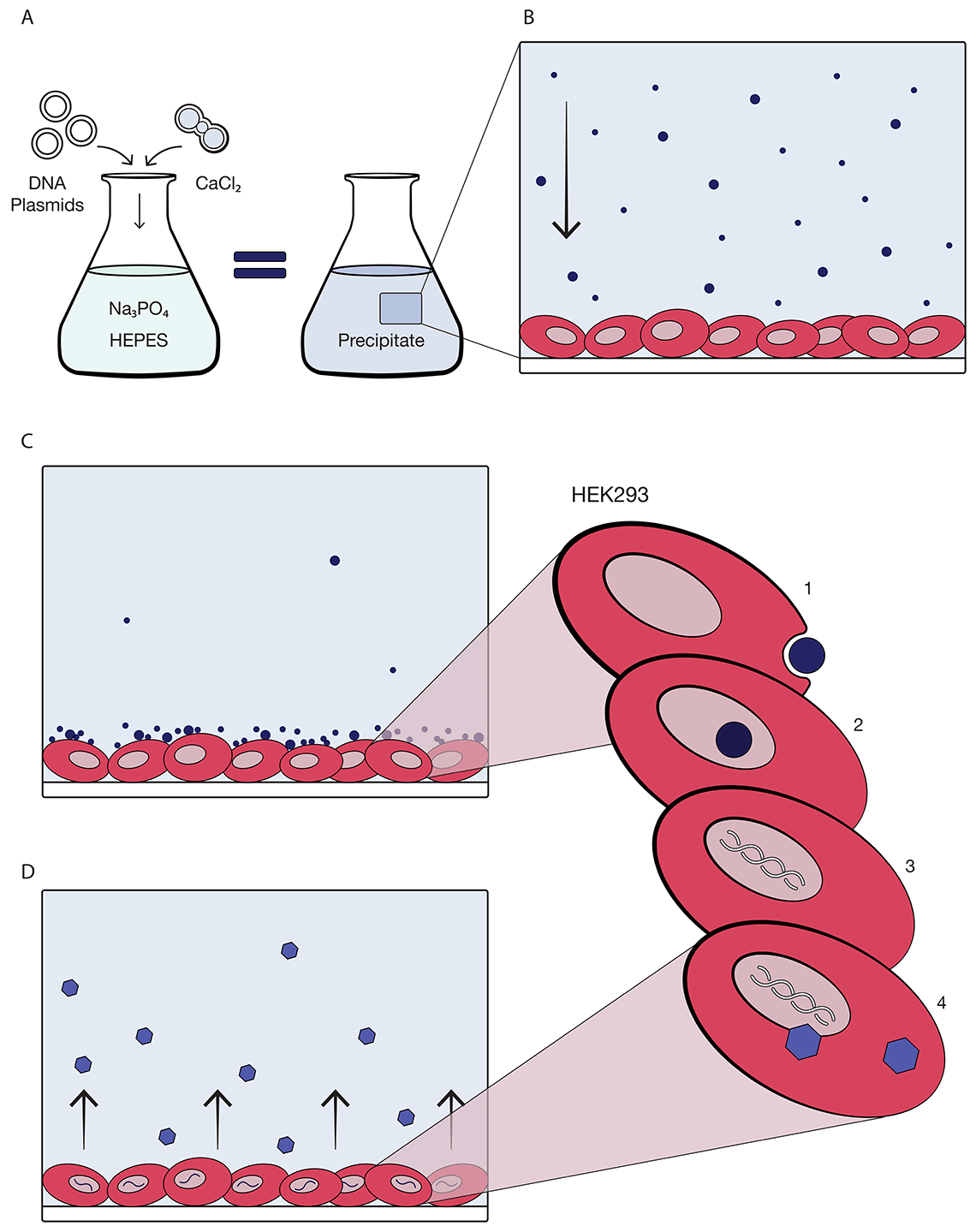
A and B: First, three DNA plasmids plus CaCl2 combine (A) with HEPES and Na2PO4 solution to form a precipitate (B) in the solution added to the HEK293 cells. C: As the precipitate settles, it is taken up by the HEK293 cell into the nucleus. Then, the cells begin processing the virus. D: The HEK293 cells assemble the virus, releasing vectors with the desired genes into the media.
Tangential Flow Filtration
Tangential Flow Filtration is a rapid and efficient method for separating and purifying the AAV vectors for the final drug product.
- Once the HEK293 cells have released the vectors into the media, the liquid is separated from the cells and filtered.
- The vector flow is parallel to the filter and is recirculated numerous times to remove large unwanted contaminating proteins.
- Diafiltration occurs to exchange the buffer and help release any vector that is retained on the filter.
- The vector is concentrated so the volume is reduced to aid in further downstream processing.
About the author
Abbie (Roth) Miller, MS, MWC, is a passionate communicator of science. As the manager of medical and science content at Nationwide Children’s Hospital, she shares stories about innovative research and discovery with audiences ranging from parents to preeminent researchers and leaders. She is a Medical Writer Certified®, credentialed by the American Medical Writers Association, and received her masters of science in Health Communication from Boston University.
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Abbie Millerhttps://pediatricsnationwide.org/author/abbie-miller/
- Posted In:
- Uncategorized




