Featured Researcher — Mingtao Zhao, DVM, PhD
Featured Researcher — Mingtao Zhao, DVM, PhD https://pediatricsnationwide.org/wp-content/themes/corpus/images/empty/thumbnail.jpg 150 150 Natalie Wilson Natalie Wilson https://pediatricsnationwide.org/wp-content/uploads/2021/06/Natalieheadshot3-2.pngMingtao Zhao, DVM, PhD, is a principal investigator in the Center for Cardiovascular Research at the Abigail Wexner Research Institute at Nationwide Children’s Hospital. He is also an assistant professor of Pediatrics at The Ohio State University College of Medicine. Dr. Zhao’s research aims to understand heart development and pioneer novel treatments for congenital heart disease using patient-derived stem cells, bringing together his interests in developmental biology and his desire to treat conditions that have affected his relatives. He and his team use genome editing tools (CRISPR/Cas9) to correct/introduce genetic variants in the context of normal and diseased heart cells originally from patients to study how they contribute to congenital heart defects.

Read on to learn more about Dr. Zhao’s work and research career.
How did you land in your field?
When I first came to the United States to pursue my doctorate degree at the University of Missouri, I studied stem cells in reproductive biology, focusing on early embryo development in mammals. I studied how an egg and a sperm were fused to form a zygote. Right after fertilization, a zygote evenly divides and becomes a two-cell embryo, which further divides to form a structure called a blastocyst, which has a big cavity inside. Embryonic stem cells (ESCs) are derived.
In 1998, the first human ESC lines were derived from blastocysts created by in vitro fertilization (IVF), but this process did not generate patient specific ESCs. In 2006, however, scientists from Japan discovered a new approach to make similar cells, induced pluripotent stem cells (iPSCs). iPSCs are like ESCs, but they don’t require human embryos for generation. They’re directly reprogramed from cells such as blood and skin cells. They can then be used to produce any cell types that make up the human body, including heart cells, brain cells, skin cells, liver cells and more. iPSCs can be patient-specific and retain all of patients’ genetic information. I was quite fascinated with iPSC technology.
By my last year of graduate school, when it came time to figure out my next steps, I had also become interested in scientific research in cardiovascular diseases and cancer, both of which had affected my family members a lot and caused them a lot of suffering. But I did not want to completely abandon what I had studied in developmental biology, and I wanted to work on real iPSCs in the dish. I pursued a postdoctoral fellowship at Stanford University, and I gradually realized that patient-specific stem cells, iPSCs, might provide the best model for studying heart disease.
Previously, human cardiomyocytes (contracting heart cells) were difficult to obtain because donated hearts were very limited, and isolated adult cardiomyocytes can only survive a few days in culture — outside the body and in a growth medium in a lab — which makes the research really challenging. Using patient-derived iPSCs, we can now generate indefinite amounts of beating heart cells in the dish. iPSC-derived cardiomyocytes are just like those found naturally in the heart and can respond to drug treatment.
Moreover, during the differentiation of iPSCs into cardiomyocytes, we can review developmental events we couldn’t study before. For most congenital heart defects, abnormalities usually start during embryonic development, but we only see diseased outcomes after birth. I think patient-specific iPSCs are great platforms to revisit and model these developmental problems and discover valuable information for therapeutic treatment.
What was your path to your current role?
I am an assistant professor (tenure-track) and direct a research lab in the Center for Cardiovascular Research as a principle investigator. It was not an easy path to this role: it took roughly nine years to reach this position, which was academically and financially challenging. I spent three years on my doctorate studies at the University of Missouri working with Dr. Randall Prather and over four years as a postdoc research fellow working with Dr. Joseph Wu at Stanford University School of Medicine. I held an instructor position for one year at Stanford Cardiovascular Institute before coming to Nationwide Children’s Hospital in August 2019.
My research has always centered around stem cell models of heart disease, particularly for congenital heart disease. I was looking for a place where I could continue that work, and I wanted to interact with clinical scientists so it could be translated from bench to bedside smoothly. I was offered a principal investigator position by Dr. Vidu Garg in the Center for Cardiovascular Research, where a number of researchers are working to understand congenital heart disease using different models and tools. My research in stem cells is unique and is complementary to the other strengths of the center.
My current studies on iPSC models of congenital heart disease perfectly match my long-time interest in developmental biology and cardiovascular disease. Bringing those two interests together is exactly what we’re doing in my lab every day. Nationwide Children’s offers a niche for me to thrive.
Fun Facts About Dr. Zhao
What would be your dream job if you didn’t work in research?
I’d like to be a computer software engineer. Computers fundamentally change the way we think and live.
What’s your favorite food?
I love Asian noodles and have eaten them since my childhood. Believe it or not, noodles have some connections with Chinese Kung Fu!
Favorite band/music?
Michael Jackson is my favorite — he’s a great dancer and singer and has influenced multiple generations around the world.
Favorite way to relax?
I love to go to the beach or mountains with my family. When I lived in California, I frequented the beach along the Pacific Ocean. I hope to spend time enjoying the Great Lakes soon when COVID-19 infections wane in the United States.
If you could have coffee with anyone (from history or who’s alive today), who would you meet?
I’d like to meet current president Joe Biden to talk to him about scientific research and ask him to invest more in it.
What is your favorite part of your role?
Every morning I come to work with full of passion and excitement because I love this job. My favorite part, though, is that I have been able to grow my lab and my research from the ground up and see it continue to move forward. Now, my lab has four members, and we’ve secured both NIH and private funding to support our research. In addition, I enjoy the research process — all of the steps from an idea to a product — though it may take multiple years to complete the mission.
How does your research serve our patients and our community?
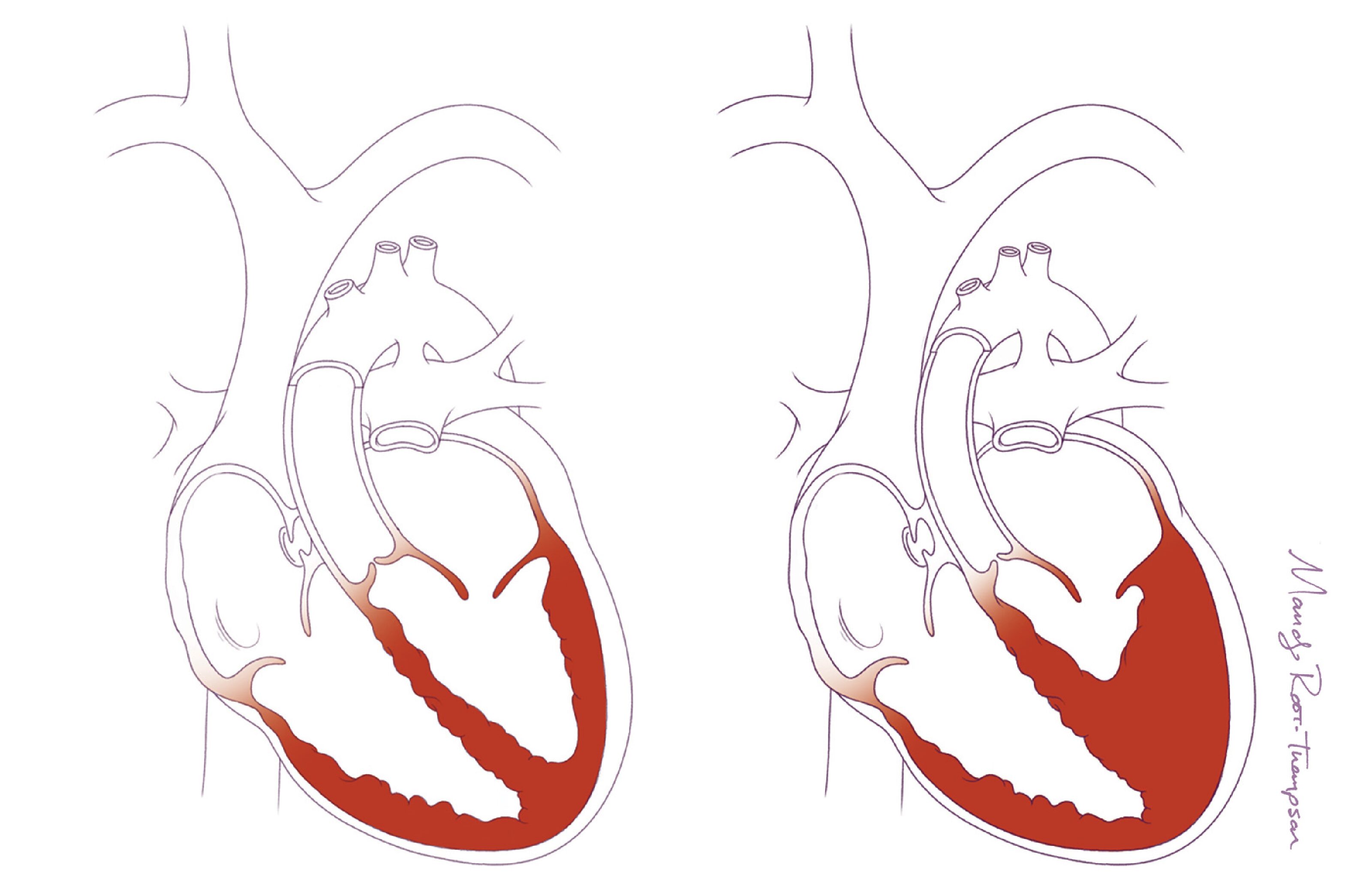
We are currently using patient-specific stem cells to study how congenital heart diseases develop. Our aim is to discover novel genes and pathways that can stimulate heart growth after birth. Some children are born with only one functional lower heart chamber (left ventricle or right ventricle). If we can stimulate the growth of the other lower heart chamber, we can significantly improve the clinical outcomes of these patients. Additionally, we hope to use stem cell-based technologies to offer personalized treatment options for our patients with severe congenital heart defects.
About the author
Natalie is a passionate and enthusiastic writer working to highlight the groundbreaking research of the incredible faculty and staff across Nationwide Children's Hospital and the Abigail Wexner Research Institute. Her work at Nationwide Children's marries her past interests and experiences with her passion for helping children thrive and a long-held scientific curiosity that dates back to competing in the Jefferson Lab Science Bowl in middle school. Natalie holds a bachelor’s degree in sociology from Wake Forest University, as well as minors in women's, gender & sexuality studies and interdisciplinary writing. As an undergraduate student, Natalie studied writing and journalism, engaged with anthropological and sociological research with a focus on race and ethnic relations, served as executive editor for the student newspaper, the Old Gold & Black, and gained marketing experience as an intern for a nonprofit entrepreneurial incubator, Winston Starts, as well as by working for Wake Forest University School of Law Office of Communication and Public Relations and its Innocence and Justice Clinic.
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Natalie Wilsonhttps://pediatricsnationwide.org/author/natalie-wilson/
- Post Tags:
- Center for Cardiovascular Research
- Posted In:
- Featured Researchers