Novel Genetic Driver Discovered for Pediatric Meningiomas Using Molecular Profiling
Novel Genetic Driver Discovered for Pediatric Meningiomas Using Molecular Profiling https://pediatricsnationwide.org/wp-content/uploads/2021/01/AdobeStock_82959563_brain-imaging-for-web-1024x575.gif 1024 575 Katie Brind'Amour, PhD, MS, CHES Katie Brind'Amour, PhD, MS, CHES https://pediatricsnationwide.org/wp-content/uploads/2021/03/Katie-B-portrait.gif
Researchers have uncovered a rare subset of meningiomas with a genetic driver shared by another cancer type, opening the door to new therapeutic considerations.
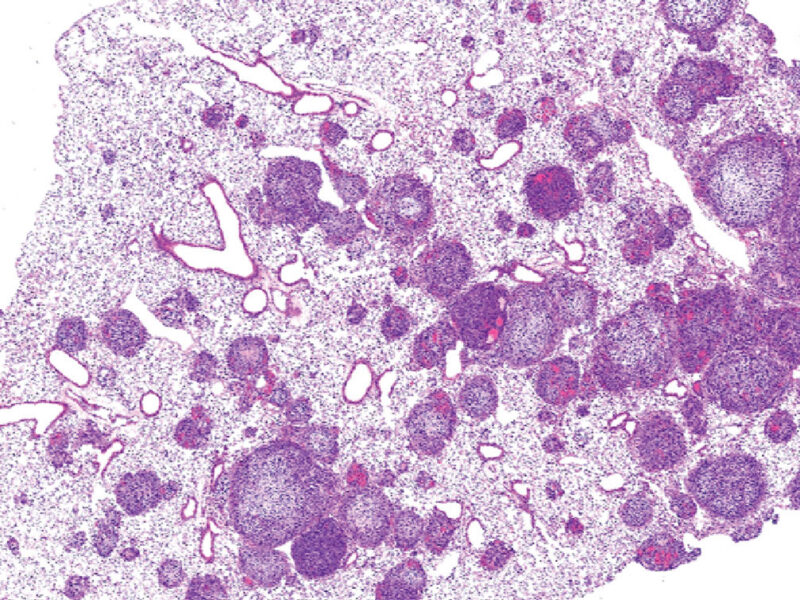
When an interesting or intractable cancer case arises at Nationwide Children’s Hospital, researchers and clinicians involved in the Brain Tumor Protocol through The Steve and Cindy Rasmussen Institute for Genomic Medicine obtain molecular profiling of the tumor in an attempt to uncover genomic information that could reveal new treatment options or generate prognostic data. When a young adult presented with an unusual meningioma, his tumor was profiled and was found to be driven by a novel and rare subtype of pediatric meningioma: an NF2-independent YAP1-FAM118B gene fusion.
Pediatric cases make up less than 1% of all meningiomas (a type of tumor that forms on the membrane of the brain or spinal cord just inside the skull), and about half of all meningiomas arise from somatic NF2 variations. The other half appear to arise from NF2-independent genetic mutations, several of which have been identified (such as SMO, AKT1 and TRAF7). When clinicians and researchers at Nationwide Children’s sent the tumor in question for genomic profiling, they uncovered a YAP1-FAM118B fusion that has otherwise been implicated only in supratentorial ependymoma.
In order to determine whether this mutation was present in other young people with meningioma, they obtained a broader set of tumor samples from 11 other non-NF2-associated meningioma patients in the Children’s Brain Tumor Tissue Consortium, an international collection of pediatric institutions involved in brain cancer research. After examining the genetic landscape of these additional tumors, they confirmed that one other child in the group had the same fusion mutation.
“This particular meningioma appears to have a similar driver to one of the subtypes of supratentorial ependymomas, but they are histologically different,” says Diana Osorio, MD, a pediatric neuro-oncologist at Nationwide Children’s and a member of the research team behind the publication, released in The American Journal of Surgical Pathology. “It would be our hope to use this type of information to learn whether similar treatment of these types of tumors produces similar results, so we can improve survival.”
Many supratentorial ependymomas and meningiomas are treated with surgical resection only, except in cases of recurrence or metastasis. Identifying therapies that effectively target this particular mutation may help guide future clinical decision-making for patients with both cancer types.
“By profiling unique or intractable tumors, we have the chance to identify existing therapies that may target the biology of specific brain tumors for which they have not yet been studied,” says Jeffrey Leonard, MD, chief of the Department of Neurosurgery at Nationwide Children’s and a member of the research team involved in the Brain Tumor Protocol. “It helps advance medical science and gives families more options when surgical resection is unsuccessful or a tumor recurs. And over time, genomic profiling will improve our understanding of tumor biology and drive up cure rates across the board.”
The Brain Tumor Protocol at Nationwide Children’s has already resulted in clinically meaningful information regarding cancer prognosis and treatment options for dozens of patients. The team aims to continue gleaning data on subtypes and genetic drivers as additional tumors are profiled, comparing them to larger samples of patient tumors from other hospitals or consortia when appropriate.
“As time progresses, the hope is that we are able to correlate what we’re seeing in the research world with whether these genetic drivers are going to be truly helpful to us clinically to result in better prognostication and therapeutics for our patients,” says Dr. Osorio. “That’s why the Institute for Genomic Medicine is so critical.”
Reference:
Schieffer KM, Agarwal V, LaHaye S, Miller KE, Koboldt DC, Lichtenberg T, Leraas K, Brennan P, Kelly BJ, Crist E, Rusin J, Finlay JL, Osorio DS, Sribnick EA, Leonard JR, Feldman A, Orr BA, Serrano J, Vasudevaraja V, Snuderl M, White P, Magrini V, Wilson RK, Mardis ER, Boué DR, Cottrell CE. YAP1-FAM118B Fusion Defines a Rare Subset of Childhood and Young Adulthood Meningiomas. American Journal of Surgical Pathology. 2020 Oct 16. Epub ahead of print.
Image credit: Adobe Stock
About the author
Katherine (Katie) Brind’Amour is a freelance medical and health science writer based in Pennsylvania. She has written about nearly every therapeutic area for patients, doctors and the general public. Dr. Brind’Amour specializes in health literacy and patient education. She completed her BS and MS degrees in Biology at Arizona State University and her PhD in Health Services Management and Policy at The Ohio State University. She is a Certified Health Education Specialist and is interested in health promotion via health programs and the communication of medical information.
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 27, 2014
- Katie Brind'Amour, PhD, MS, CHEShttps://pediatricsnationwide.org/author/katie-brindamour-phd-ms-ches/April 28, 2014