Signaling Pathway Changes May Flag CAVD, Offer Target for Therapies
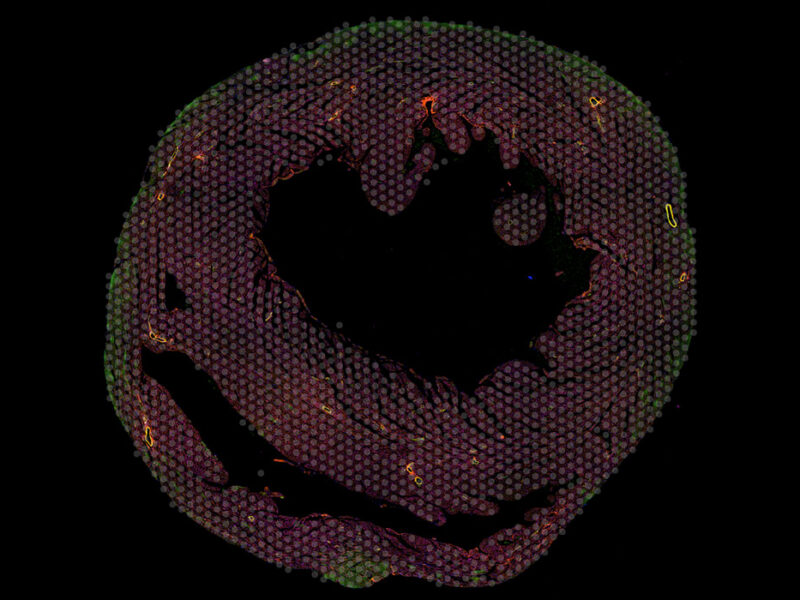
Signaling Pathway Changes May Flag CAVD, Offer Target for Therapies https://pediatricsnationwide.org/wp-content/themes/corpus/images/empty/thumbnail.jpg 150 150 Kevin Mayhood Kevin Mayhood https://secure.gravatar.com/avatar/5b2c3505cd7c7ea0367bd2da6478c8e246b6f4511092fa0e98606f4eaaa6d1e0?s=96&d=mm&r=gLoss of TgfB1 from the endothelium leads to a reduction in Sox9 expression and valve calcification.
A team of researchers has identified a molecular signaling pathway that, when altered, can contribute to calcific aortic valve disease (CAVD). The finding may provide a method for early diagnosis — many patients don’t learn they have the disease until it’s in the final stage — and a target for treatment therapies.
CAVD is the most prevalent valvular disorder in the United States, sending 55,000 patients to the hospital and causing 15,000 deaths annually. Long thought to be acquired during adulthood, there is increasing evidence to suggest the disease has its origins during embryonic development.
“The only effective therapy available now is surgical repair and replacement, which is very expensive and a very high risk burden to the patient,” says Joy Lincoln, PhD, a principal investigator in the Center for Cardiovascular Research at The Research Institute at Nationwide Children’s Hospital and leader of the study.
“Surgical intervention costs the U.S. government around $2 billion per year,” says Dr. Lincoln, who is also an associate professor of Pediatrics at The Ohio State University College of Medicine. “A less costly and less risky alternative would be beneficial to affected individuals.”
Building on her earlier work that found a reduction of the transcription factor Sox9 resulted in valve calcification in mice, Dr. Lincoln and her colleagues focused on identifying the signaling pathways that regulate this process.
“We and others have shown that calcification of the valve is similar to bone development,” she says.
In mouse, porcine and human in vitro assays of isolated valve cells, they found that TgfB1 is secreted by the layer of endothelial cells that overlie the aortic valve and molecularly communicate with underlying valve interstitial cells to regulate Sox9 expression. In mice, loss of TgfB1 in the valve endothelium leads to a reduction in Sox9 expression in interstitial cells and subsequent CAVD.
“TgfB1 promotes calcification in the bone,” Dr. Lincoln says. “But, in our system, it had the opposite effect, which was surprising to us.”
She believes that changes found in the signaling pathway could possibly be used to help identify molecular landmarks of CAVD, leading to earlier diagnoses.
The researchers are now investigating how genetic, environmental and biomechanical factors may trigger or influence the calcification process and whether they can manipulate signaling pathways or cell function to either reverse the disease or stop it from getting worse.
Reference:
Huk DJ, Austin BF, Horne TE, Hinton RB, Ray WC, Heistad DD, Lincoln J. Valve endothelial cell-derived TGFB1 signaling promotes nuclear nuclear localization of Sox9 in interstitial cells associated with attenuated calcification. Arteriosclerosis, Thrombosis and Vascular Biology. 2016 Feb;36(2):328-38.
About the author
- Kevin Mayhoodhttps://pediatricsnationwide.org/author/kevin-mayhood/
- Kevin Mayhoodhttps://pediatricsnationwide.org/author/kevin-mayhood/April 25, 2015
- Kevin Mayhoodhttps://pediatricsnationwide.org/author/kevin-mayhood/April 25, 2015
- Kevin Mayhoodhttps://pediatricsnationwide.org/author/kevin-mayhood/April 25, 2015
- Posted In:
- In Brief