Accelerating Pediatric Medical Device Innovation
Accelerating Pediatric Medical Device Innovation https://pediatricsnationwide.org/wp-content/themes/corpus/images/empty/thumbnail.jpg 150 150 Erin Gregory Erin Gregory https://secure.gravatar.com/avatar/?s=96&d=mm&r=gThe Midwest Pediatric Device Consortium (MPDC) is a unique collective led by Nationwide Children’s Hospital and The Ohio State University, designed to facilitate the development, production, and distribution of pediatric medical devices. Comprising a diverse group of leaders in universities, healthcare systems, community organizations, and industry partners across Ohio, the MPDC is committed to transforming ideas into practical solutions for pediatric medicine.
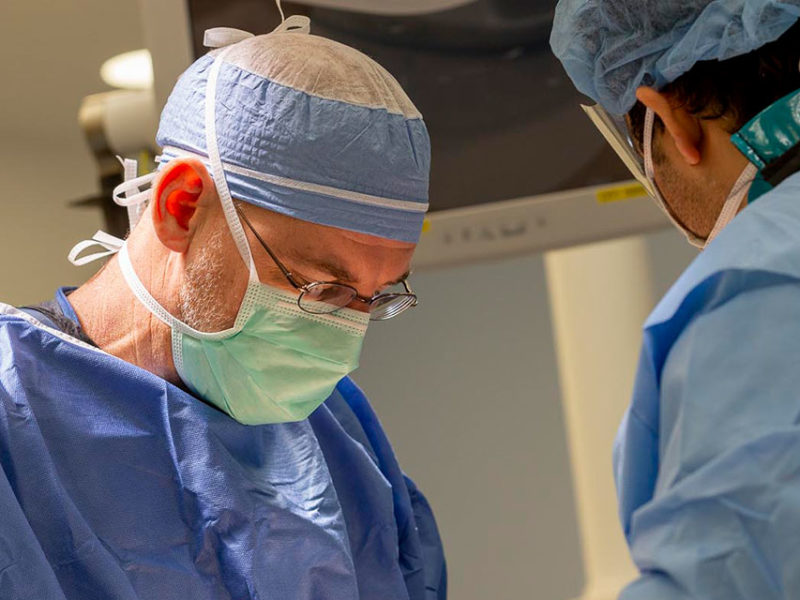
The MPDC is supported by a $6.95 million grant from the Food and Drug Administration (FDA) awarded to co-principal investigators Cory Criss, MD, (pictured below, right) chief operating officer of the consortium and pediatric surgeon at Nationwide Children’s Hospital, and David Eckmann, PhD, MD, (pictured below, left) chief executive officer of the consortium and founding director of the Center for Medical and Engineering Innovation (CMEI) at The Ohio State University College of Medicine.
The grant’s primary priority requirement is to bring pediatric medical devices into the marketplace. This initiative plays a pivotal role in advancing pediatric medical device development throughout the United States. MPDC, in conjunction with other Pediatric Device Consortia (PDCs), not only provides funding but also offers invaluable mentorship, transforming inventors into successful innovators.

What Are the Mission and Vision of the MPDC?
The MPDC’s strength lies in its collaborative approach, with members representing premier institutions including Nationwide Children’s, Ohio State, Cleveland Clinic Children’s, Cincinnati Children’s, and other health care centers, academic institutions, and industry partners across the Midwest.
“This collaboration encourages a positive exchange of knowledge and resources, positioning us to swiftly advance innovative ideas into pediatric devices available across the nation,” says Dr. Eckmann. “We are driven by a common vision: to leverage the expertise of our consortium to expedite the development of pediatric medical devices. This vision is rooted in the belief that every child deserves access to the most advanced and effective medical technologies.”
What Sets the MPDC Apart?
“Our academic/industry hybrid model focuses on commercialization, regulatory navigation, trial implementation, individualized mentorship, and a commitment to diversity, equity, and inclusion,” says Dr. Criss. “This is a unique approach to pediatric device development.”
The dedicated consortium-member experts provide individualized support, ensuring that innovators’ ideas are brought to life.
“Our reliance on commercialization, industry partnership, and venture funding makes us a significant contributor to the PDC network,” says Dr. Eckmann.
How Do Projects Get Funded?
To support clinicians, researchers, and inventors in their quest for pediatric medical device innovation, MPDC offers a formal application process that will be available through its website. The application processes across the various PDCs are not geographically restricted, and collaboration among PDCs is encouraged to collectively advance pediatric device development.
Funding processes may vary from one consortium to another, but typically involve application submission, screening and review, and opportunities for pitching ideas. Beyond funding, MPDC is committed to mentoring, educating, and counseling innovators to help them move forward, whether it’s for the next funding cycle or other avenues of product development.
Who Else Is Part of the MPDC?
The driving force behind MPDC includes primary participants such as Nationwide Children’s Hospital, The Ohio State University, Cleveland Clinic Children’s, and Cincinnati Children’s Hospital Medical Center. Additionally, Ohio Life Sciences, Rev1 Ventures, and many other organizations play a pivotal role. Innovators who submit their ideas for evaluation are essential contributors, as the MPDC thrives on their alliance.
About the author
- Erin Gregoryhttps://pediatricsnationwide.org/author/erin-gregory/September 27, 2023
- Erin Gregoryhttps://pediatricsnationwide.org/author/erin-gregory/
- Erin Gregoryhttps://pediatricsnationwide.org/author/erin-gregory/
- Erin Gregoryhttps://pediatricsnationwide.org/author/erin-gregory/