Biofilm Bacteria Protected by a Form of DNA Resistant to Current Treatments
Biofilm Bacteria Protected by a Form of DNA Resistant to Current Treatments https://pediatricsnationwide.org/wp-content/themes/corpus/images/empty/thumbnail.jpg 150 150 Lauren Bakaletz, PhD and Steve Goodman, PhD Lauren Bakaletz, PhD and Steve Goodman, PhD https://pediatricsnationwide.org/wp-content/uploads/2016/04/LaurenBakaletz.jpgResearchers from Nationwide Children’s Hospital identify a structural component of the bacterial biofilm matrix in a novel discovery published in Cell that has strong implications as to how biofilms resist current treatments and how to create new therapeutics that will be effective.
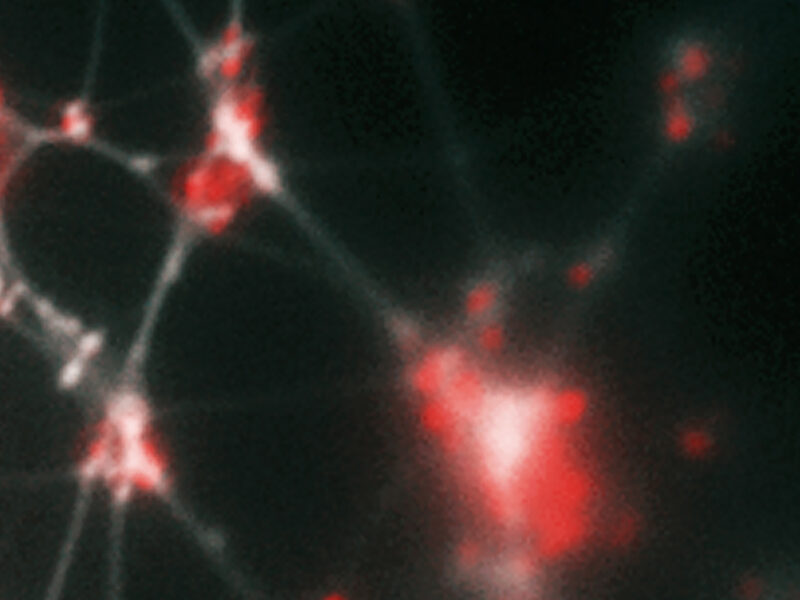
Biofilms are communities of bacteria that are protected by an extracellular matrix that is rich in and requires DNA. When present in the body, biofilms act as the fortresses that shield bacteria from antibiotics and immune responses. While enzymes that digest DNA — for example, DNase and Pulmozyme® — can prevent biofilm formation, these same enzymes fail to disrupt biofilms that are mature despite the fact that extracellular DNA (eDNA) continues to accumulate within the biofilm over time.
This conundrum has baffled researchers in the biofilm field and clinicians who treat biofilm mediated diseases such as the chronic, and ultimately fatal, pulmonary infections associated with cystic fibrosis. In these cases, treating with DNase has minimal efficacy.
Extracellular DNA (eDNA) is a key component of biofilm matrix structure. In a new study published in Cell, researchers from Nationwide Children’s Center for Microbial Pathogenesis present evidence that this abundant eDNA is not the typical well-known B-form of DNA, but is, instead, the rare Z-form of DNA.
Z-DNA forms a left-handed double helix, unlike the more common B-DNA, which forms a right-handed helix. Also, unlike B-DNA, a biological role for Z-DNA was not obvious.
Hallmarks of the Z-DNA structure include its left-handed double helix, zig zag appearance, increased stiffness compared to B-DNA and its resistance to all known enzymes (nucleases) that degrade B-DNA. While Z-DNA is associated with gene regulation, innate immune sensing and inflammation, its appearance is uncommon and typically fleeting.
This new study shows not only that Z-DNA in the biofilm is stable and the more structurally important form of eDNA in biofilms than B-DNA but also that it confers eDNA, and thus the biofilm matrix, with the empowering nuclease resistant property.
We found that not only does the Z-DNA matrix protect the bacterial biofilm, it also acts offensively to inactivate the part of the immune system that restricts biofilm growth. This two-pronged effect has strong implications for treating biofilm infections and other autoimmune diseases such as systemic lupus erythematosus (aka Lupus), where the counter-effective immune response drives Z-DNA formation and thus biofilm growth.
To test the hypothesis that Z-DNA was the main component of biofilm eDNA, our team conducted a series of experiments showing the following:
- Z-DNA was present in all tested biofilms, both in vitro and in vivo
- Z-DNA was the primary structural form of eDNA within established biofilms
- Z-DNA antibodies stimulated biofilm formation
- Shifting the ratio of B- to Z-form DNA changed the biofilm structure
In previous studies the team demonstrated that eDNA was important throughout the course of a biofilm infection. In this study, the goal was to answer the burning question of why this eDNA became resistant to DNases.
Biofilms are an ancient means for bacteria to protect themselves against harsh environments. In order for different species of bacteria to work together in a biofilm they need to have the same materials for, and to know how to build, a compatible fortress. The term biofilm was coined by Bill Costerton in 1978. One year later Z-DNA was discovered. We never would have imagined the two fields would intersect so dramatically when we started this project.
Now, with an understanding of the role of Z-DNA in the eDNA matrix, diseases that were untreatable can be examined with new eyes. Z-DNA is not just part of the biofilm, it impinges on the immune system in a way that begins to tie a lot of loose ends together. Developing therapeutic agents to drive the biofilm eDNA from the Z-form back to the native B-form could provide additional approaches for clinical treatment or prevention of biofilm-mediated diseases.
This work not only directly leads us into the clinic but also explains how microorganisms and the host immune system co-evolved. With this new understanding as to why DNase resistance of biofilms occurs, a big domino has fallen, and we anticipate that others in host-pathogen interrelationship will follow.
Reference:
Buzzo JR, Devaraj A, Gloag ES, Jurcisek JA, Robledo-Avila F, Kesler T, Wilbanks K, Mashburn-Warren L, Balu S, Wickham J, Novotny LA, Stoodley P, Bakaletz LO, Goodman SD. Z-form extracellular DNA is a structural component of the bacterial biofilm matrix. Cell. 2021 Nov 3. [Epub ahead of print]
About the author
Lauren Bakaletz, PhD, is the director of the Center for Microbial Pathogenesis, Tillie E. Coleman Endowed Chair in Pediatric Research and vice president for Basic Sciences Research at Nationwide Children’s Hospital. She is also the recipient of the
2021 Steve Allen Distinguished Scholar Award.
-
Steve Goodman, PhDhttps://pediatricsnationwide.org/author/steve-goodman-phd/