How to Solve Feeding Disorders Without a G-Tube
How to Solve Feeding Disorders Without a G-Tube https://pediatricsnationwide.org/wp-content/uploads/2017/04/021317ds0204G-cover-story-header-1024x575.gif 1024 575 Jeb Phillips Jeb Phillips https://pediatricsnationwide.org/wp-content/uploads/2021/03/Jeb-Phillips.jpg- April 21, 2017
- Jeb Phillips

Babies in a neonatal intensive care unit must have a safe way of receiving nutrition in order to go home. Full oral feeding is ideal, of course. But for those patients with persistent difficulty feeding by mouth, there were two primary options before 2002 to guarantee nutrients by the time of discharge.
Both had their own problems.
Option One. An infant remained an inpatient while specialists and therapists tried empiric therapy that could lead to effective feeding. Maybe an acid suppressive medication would do the trick. Maybe formula thickeners would. Few objective tests could tell providers what was underlying the baby’s feeding difficulties, so doctors made educated guesses based on clinical symptoms.
Extending inpatient stays until a baby achieves oral feeds might have negative health consequences, it’s expensive, and there was no way to predict how long the stay would be. In an era of medical cost containment, the first option is not much of one.
Option Two. Send the infant home eating by tube – often meaning the placement of a gastrostomy tube (G-tube) to deliver nutrition directly to the stomach. The maintenance of these tubes is associated with its own costs. They add to new parents’ already considerable stress, and G-tube complications can send infants back for an inpatient stay. Crucially, a G-tube bypasses all the structures above the stomach, eliminating opportunities for a baby to develop oromotor, airway protection and related higher neural functions.
A 2016 study found that the presence of G-tube at discharge from a NICU was an independent predictor of neurodevelopmental delay at 2 years of age.
There is now a third option. Largely developed by Sudarshan Jadcherla, MD, director of the Neonatal and Infant Feeding Disorders Program at Nationwide Children’s Hospital, it was 20+ years in the making. The majority of babies referred to the Nationwide Children’s program since 2008 for placement of a G-tube are actually feeding by mouth at discharge.
“Not everyone can do exactly what we do, but people need to know that it is possible,” says Dr. Jadcherla. “Survival of premature infants, of high-risk infants, is increasing. That is important on its own. We also need to focus on survival without morbidity, though. Our overarching, long-term vision with our feeding interventions is to improve the life of the child.”
When Does a Feeding Disorder Begin?
Dr. Jadcherla, now the associate division chief of Neonatology at Nationwide Children’s, started his pediatrics and pediatric gastroenterology training in Ireland, caring for children who had long-standing eating disorders.
The underlying causes were gastroenterological, neurological or cardio-respiratory, and as the child aged, behavioral components of feeding difficulties emerged. Successful therapies become more difficult to implement when all four are in play.
So Dr. Jadcherla decided to back up. He wanted to know how the neuro-gastroenterological system developed during gestation and early infancy. How does normal peristalsis even occur for a baby? How does airway protection function? What are the physiological and pathophysiological bases for airway and digestive symptoms? With answers to these and other questions, can we learn how to treat feeding issues in the neonatal time period before they become entrenched?
An early 1990s neonatology fellowship focusing on GI motility at Baylor College of Medicine, and some emerging research into neurological, airway and digestive relationships were foundational. Airways and the esophagus have common innervation, this research was showing. The gut originates early in the embryological stage, and airway and lung buds arise from that primitive gut. Stimulating the foregut means the airway and lungs can react, and vice versa.
Clinicians and researchers at the Medical College of Wisconsin, Milwaukee were pioneers in developing objective evaluations for adult swallowing and feeding disorders, including esophageal manometry. Dr. Jadcherla became a faculty member at the college and the affiliated Children’s Hospital of Wisconsin in 1995, and began working to apply some adult ideas to neonates.
Among their innovations, Dr. Jadcherla and others in Wisconsin designed manometry catheters to measure esophageal motility in babies as small as 0.7 kilograms (1.5 pounds).

“At this point, I was really just trying to understand what normal looks like for infants,” says Dr. Jadcherla. “We cannot objectively know what is abnormal until we understand which esophageal reflexes ensure aerodigestive protection in normal infants, or until we characterize the sensory-motor properties of these reflexes during maturation.”
In 2002, Dr. Jadcherla moved to Nationwide Children’s.
Characterizing Abnormal
The study describing the creation of the infant-sized manometry catheter did not appear until 2003. So Dr. Jadcherla was bringing unpublished possibilities with him on his move to Ohio.
“I started working in the NICU two years before Dr. Jadcherla arrived,” says Rebecca Moore, RN, and clinical research coordinator in the Innovative Feeding Disorders Research Program at Nationwide Children’s. “There were no manometry studies here or anywhere else that I knew of. We didn’t have many diagnostic tools that allowed us to see what feeding abilities a baby had. We only had clinical observation. A lot of babies were going home with G-tubes.”
That started to change as Dr. Jadcherla continued gathering normative data at Nationwide Children’s, but shifted part of his focus to characterizing abnormality in feeding. A series of studies, abstracts and presentations reported on investigations into how pharyngo-esophageal reflexes are impaired with perinatal neurological injury, in the presence of gastroesophageal reflux disease (GERD) and in infants with bronchopulmonary dysplasia (BPD). Over the next few years, Dr. Jadcherla started formulating what was beginning to look like that third option – somewhere between an indefinite stay in the NICU and a rush to place a G-tube so a neonate could be discharged. Research from him and others suggested that a “pathophysiology-based approach” to feeding disorders was both possible and useful.
The techniques for evaluation that Dr. Jadcherla had helped create would allow specialists to map the suckswallow-peristalsis process of an infant. The normative data he had collected would allow the abnormalities to be pinpointed: Is the issue reflux, coordination of esophageal muscles, aspiration? A combination, or something else entirely? Then a personalized therapy strategy could target the exact problems.
Dr. Jadcherla fully introduced the Neonatal and Infant Feeding Disorders Program in 2008.
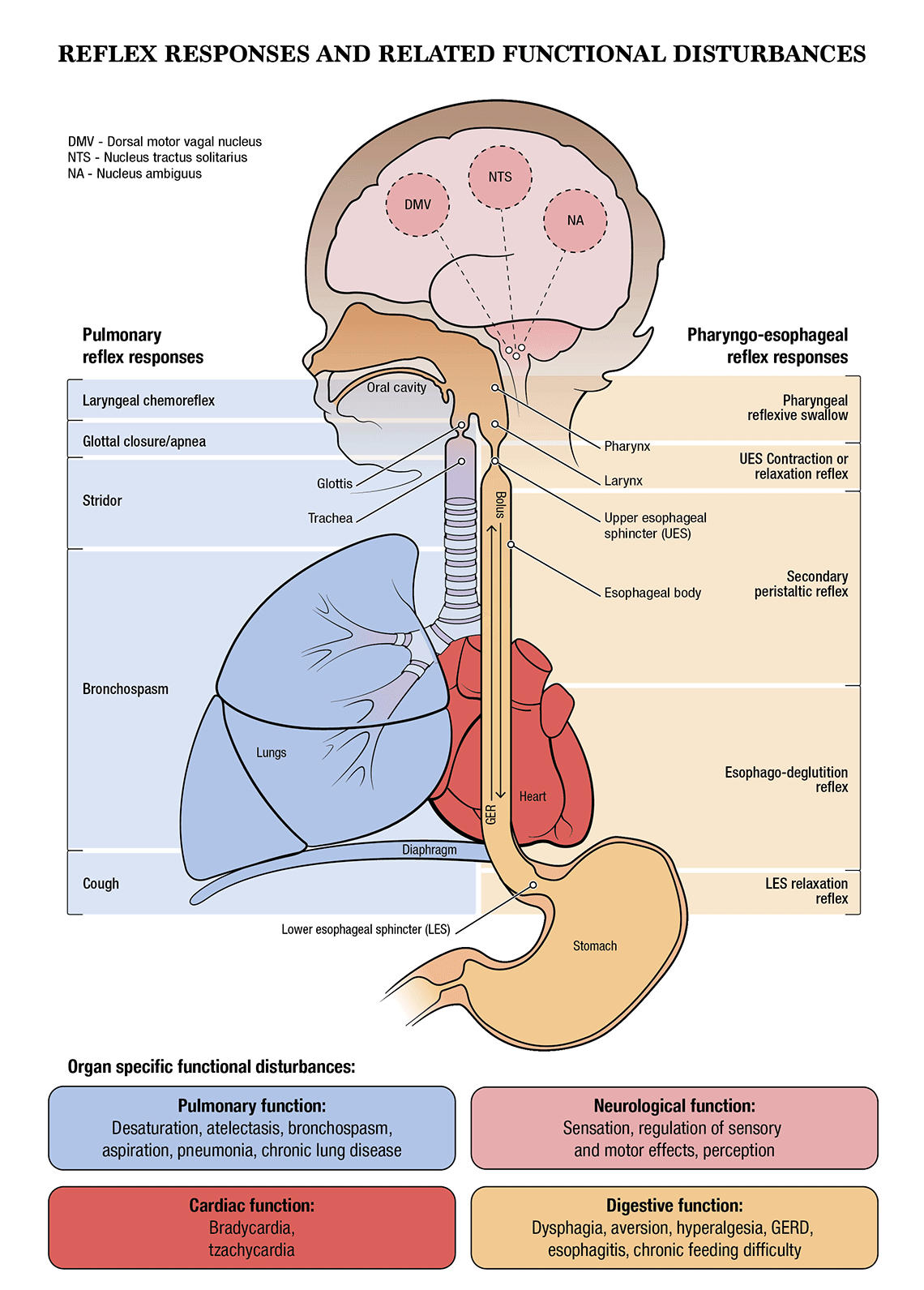
Precision Medicine at the Cribside
When a baby is referred from the NICU with feeding problems severe enough that a G-tube may be warranted for discharge, program staff members perform a complete clinical evaluation. Special attention is paid to the volume fed, the type of feed and frequency of feeding. It’s important to have a clear picture of all the feeding milestones during the patient’s life, not just the current feeding situation, Dr. Jadcherla says.
Other, previously diagnosed conditions are also considered. Neurological impairment, lung disease and airway issues all may affect the neuro-aerodigestive apparatus. A videofluoroscopic swallow study or upper GI fluoroscopy study may have already been performed, and those become part of the evaluation. If they haven’t been performed, they typically aren’t unless a structural abnormality is suspected.
A pH impedance study is often the first diagnostic tool used for an infant referred to the program. Nearly all babies experience reflux, and GERD is a frequent clinical diagnosis when irritability, lack of weight gain or other symptoms become troublesome. The impedance study determines how acidic the refluxate is, how high it travels up the esophagus and how long it stays before a baby can clear it. A diagnosis of GERD is often made based on symptoms. These impedance studies regularly show that GERD is not the primary problem.
“GERD is diagnosed more often than it should be,” says Dr. Jadcherla. “Reflux is common and can result in symptoms. Many symptoms in a premature baby appear to be troublesome because the baby has little tolerance for any symptoms. The feeding disorders themselves often have other causes.”
The most in-depth study is swallow-integrated esophageal manometry with concurrent monitoring of respiration patterns, heart rate patterns and pulse oximetry. Better than any other evaluative tool currently available, this allows Dr. Jadcherla and his team to understand what happens after a bolus is introduced into the oral cavity – length and frequency of muscle contractions, reactivity to a stimulus, tone of upper and lower esophageal sphincters
and, if the swallow stops before completion, where it stops.
“With this study, we are in a position to understand oral mechanics like suction and expression and peristalsis across the pharynx and esophagus,” Dr. Jadcherla says. “At the same time, we are studying airway function and heart function. We can discover where the symptoms are being caused, and we can make a treatment plan.”
Sometimes, the best plan after the evaluation ends up being a G-tube anyway.
Dr. Jadcherla and colleagues have discovered and characterized important manometric markers that are associated with feeding success:
- Sphincteric response to esophageal and pharyngeal infusion
- Peristalsis induced by esophageal and pharyngeal infusion
- Secondary peristalsis induced by esophageal infusion
- Normal pharyngeal manometry
- Oral feeding challenge success
- Suck-swallow-breathe-esophageal swallow sequence
The absence of one or two of these does not mean feeding therapy will fail. The absence of most, though, suggests that a baby is not developmentally mature enough for therapies to work. A better use of time and resources would be to place a G-tube and allow the baby to grow until therapies have a better chance of making a difference, says Dr. Jadcherla.
“That can be disappointing for parents, but because we have always included parents in this decision-making process, they understand why we believe a G-tube is the appropriate option at that point,” says Dr. Jadcherla. “A G-tube is not the end of our commitment to these families. In some ways, it represents the beginning. For some infants, rehabilitation cannot start until they mature and have stable breathing patterns.”
When manometry and other evaluations indicate the potential for success without a G-tube, though, the feeding team begins plotting a strategy.
“Repetition is the Mother of Skill”
Pharmacological treatment may be indicated in some circumstances. An acid-suppressive medication is given if the team objectively determines that reflux is abnormally acidic and is a cause of difficulties. Infants with BPD, and neurological or cardiac conditions, may have other drugs that are indicated for their diseases.
Therapy in the Neonatal and Feeding Disorders Program largely entails taking the time to fine-tune how the baby is fed by mouth and train the baby’s many sucking-feeding-swallowing reflexes. That means, in the first place, adjusting volume and frequency of feeds, viscosity, nipple flow rates and caloric density. In the second, it means a variety of occupational therapy tactics, including cue-based feeds, pacing of feeding, posture and operant conditioning.
“Repetition is the mother of skill,” Dr. Jadcherla explains. “A very small quantity of oral feed can help train the neuromuscular apparatus. Even 5 mL provides the infant with sensation – taste, liquidity. The stimuli activate different neural pathways resulting in peristalsis. You must repeat these therapies to help the baby develop the neuro-aerodigestive interrelationship. You cannot try one thing, then move on to the next, without giving the baby a chance to respond.”
A baby’s behavioral state matters, Dr. Jadcherla says. A child using neuromuscular resources to cry is not going to focus on oral skills. A calm, bundled baby who receives eye contact from the feeder will learn more quickly. A baby continually fed by gavage tube does not experience hunger. Hunger is a prelude to the act of feeding, so therapists adjust how often feeding occurs.
A baby fed by gavage tube has also not learned to associate sucking with nutrition or the feeling of fullness. So when a tube feed does take place, a therapist may have the infant suck on a pacifier simultaneously. Sucking on a pacifier alone does not always provoke swallowing, so a small stimulus like water, milk or sucrose is included. A baby then knows that hunger, sucking, swallowing and feeling full are connected.
“Physicians and therapists can be afraid to start oral feeds, especially when infants remain on oxygen,” says Moore, the research program coordinator. “We know from our own experience, and from our research, that it is developmentally appropriate, and actually good for development.”
Baby Paul
There are now nearly 100 peer-reviewed publications supporting the methods of the Neonatal and Infant Feeding Disorders Program. There is also 8-month-old Paul Terlecky.
Paul’s mother, Rachel, is an emergency medicine physician at Licking County Memorial Hospital in Newark, Ohio, and his father, Andrew, is an orthopedic surgeon there. The couple took a 2016 trip to Hawaii when Rachel was pregnant. Just before they were scheduled to come home, Rachel went into labor.
Paul was born at 31 weeks gestational age and spent 8 weeks in the NICU at Kapiolani Medical Center for Women and Children before he was stable enough to travel home. He was fed by nasogastric tube until a week or so before discharge. Paul had episodes of cyanosis during bottle feeding, but breast feeding was going well, Rachel says.
Six days after the return to Ohio, she noticed that he was having more severe episodes of cyanosis. They were now occurring during breast feeding. A pulse oximeter showed Paul’s SPO₂ levels were dropping below 80 percent during those episodes. The Terleckys went to the Nationwide Children’s Emergency Department, where Paul was admitted.
Because of Paul’s age and symptoms, he was referred to Dr. Jadcherla and the feeding program. Paul underwent manometry, which showed that his nasopharynx and upper esophageal sphincter were hyper-reactive to stimulus. In essence, the reflexes that exist to protect the airway were doing too good of a job. Any tiny liquid volume was causing him to gag and cough. A fiberoptic endoscopic evaluation of swallowing (FEES) showed he was usually not aspirating with feeding.
Why the hyper-reaction now, after a period of relatively safe feeding? Lactation consultants weighed Paul before and after a short breastfeeding session and found that he was gaining an ounce in a just a few minutes. Rachel, it turned out, had a fast let-down and was providing more milk than Paul could safely swallow in the week or so since he left the NICU in Hawaii. That likely led to Paul’s protective mechanisms becoming overprotective.
Dr. Jadcherla told the Terleckys that therapy could remedy this, and he led a care conference with the parents laying out the plan. Rachel Terlecky becomes emotional when talking about it.
“He took the time to connect with his patients, to make sure we felt heard and to make sure our objectives were met. I cannot say enough about the way he cared for us as a person and as a physician.”
Paul took some thickened feeds from a bottle. The lactation team worked with Rachel to safely decrease her supply, including having her pump off milk before breast feeding. Paul would eat in a side-lying position, so that he (and not gravity) could control the amount of milk he was swallowing. He was discharged after nine days, and side-lying is the only modification the Terleckys still use.
“We know there are children who have G-tubes and ongoing problems,” Rachel says. “I think those kids are lucky that they have Dr. Jadcherla.”
The Future
What about the neonatal populations of other states or other countries?
“Institutions are starting to replicate programs like the one at Nationwide Children’s Hospital and programs for older children,” says Neelesh Tipnis, MD, the chair of Pediatric Gastroenterology at the University of Mississippi Medical Center who began a similar program at that institution three years ago. “When I came here, we were the fourth in the South. There are now eight pediatric motility programs. People recognize the importance of these skilled, interdisciplinary teams and how important they are in helping children reach their full potential.”
Recognition, however, comes with the realization that a lot of work still must be done, says Dr. Tipnis. Dr. Jadcherla’s research in the youngest population is crucial, because it provides a starting point for learning how children’s feeding disorders change as they age. Significant knowledge gaps still exist across the pediatric age spectrum, however. Dr. Tipnis often first sees patients after they leave the NICU.
“Our techniques are getting better, our ability to analyze big data is getting better,” Dr. Tipnis says. “We still do not have much normative data in older children, though. From a practical standpoint, as reimbursement continues to change, we are not going to be compensated for treatments that don’t have objective evidence behind them.”
The continuing lack of objective evidence – along with a lack of professionals experienced in interpreting evaluations and treating patients – also worries Joan Arvedson, PhD, program coordinator of Feeding and Swallowing Services at Children’s Hospital of Wisconsin-Milwaukee. Dr. Arvedson is co-author of Pediatric Swallowing and Feeding: Assessment and Management, one of the field’s best-known reference books.
“The standardized measures that Dr. Jadcherla and his colleagues gather are continuing to build toward a stronger clinical basis for treatment,” she says. “I have great concerns about the variability of interpretation that still exists, particularly in videofluoroscopic swallow studies. I have concerns in some cases about the limited evidence for what some clinicians and therapists do. We must have appropriate tests, but we also need professionals to know what they are doing with those tests.”
Dr. Arvedson and Dr. Jadcherla agree, in her words, that “the most developmentally, neurophysiologically appropriate therapies are what we should be focused on.” Dr. Jadcherla and his colleagues at Nationwide Children’s continue to caution neonatologists and pediatric gastroenterologists about the use of acid suppressive medications, when there is mounting evidence that they are often unhelpful and may harm some children. Dr. Arvedson says that in her experience, formula thickeners are too frequently employed as the first change made, and they may have negative consequences for the GI tract or even increase risk for aspiration.
Dr. Jadcherla says one of the larger concerns in neonatal feeding disorders is how smaller institutions, or physicians in developing countries, can use these cutting-edge techniques and research to help their patients. Neonatal esophageal manometry, for example, is only possible in a few major research institutions.
“We need a multicenter trial to come up with simplified criteria,” he says. “We can disseminate the knowledge we have now, so people can at least try to make educated guesses. But can our methods be used in the absence of the resources only available at a few institutions? For example, if an institution does not have the resources to do any objective evaluations, and a neonate with a feeding disorder is likely to go home with a G-tube, can we at least give an optimal post-menstrual age for G-tube placement?”
Nationwide Children’s has now embarked on a single center, randomized, controlled trial to investigate diagnosis and treatment of GERD in infants. Dr. Jadcherla continues to study the mechanisms of aspiration during swallowing and how aspiration can be rectified.
And day to day, he sees children like Paul who can learn to feed well when given personalized treatment. “Our work to improve the quality of these babies’ lives continues,” Dr. Jadcherla says.
References:
- Jadcherla SR, Khot T, Moore R, Malkar M, Gulati IK, Slaughter JL. Feeding methods at discharge predict long-term feeding and neurodevelopmental outcomes in preterm infants referred for gastrostomy evaluation. The Journal of Pediatrics. 2017 Feb; 181:125-130.
- Jadcherla SR, Peng J, Moore R, Saavedra J, Shepherd E, Fernandez S, Erdman SH, DiLorenzo C. Impact of personalized feeding program in 100 NICU infants: pathophysiology-based approach for better outcomes. Journal of Pediatric Gastroenterology and Nutrition. 2012 Jan; 54(1):62-70.
- Jadcherla SR, Stoner E, Gupta A, Bates DG, Fernandez S, Di Lorenzo C, Linscheid T. Evaluation and management of neonatal dysphagia: impact of pharyngoesophageal motility studies and multidisciplinary feeding strategy. Journal of Pediatric Gastroenterology and Nutrition. 2009 Feb; 48(2):186-192.
- Jadcherla SR, Duong HQ, Hoffmann RG, Shaker R. Esophageal body and upper esophageal sphincter motor responses to esophageal provocation during maturation in preterm newborns. The Journal of Pediatrics. 2003 Jul; 143(1):31-38.
- Jadcherla SR and Rudolph CD. Gastroesophageal reflux in the preterm neonate. NeoReviews. 2005 Feb;6(2): e87-e98.
- Jadcherla SR, Hasenstab KA, Sitaram S, Clouse BJ, Slaughter JL, Shaker R. Effect of nasal noninvasive respiratory support methods on pharyngeal provocation-induced aerodigestive reflexes in infants. The American Journal of Physiology – Gastrointestinal and Liver Physiology. 2016 Jun 1; 310(11):G1006-G1014.
Image credits: Nationwide Children’s; photos of newborn baby Paul courtesy of Rachel and Andrew Terlecky
About the author
Jeb is the Managing Editor, Executive Communications, in the Department of Marketing and Public Relations at Nationwide Children's Hospital. He contributes feature stories and research news to PediatricsOnline, the hospital’s electronic newsletter for physicians and other health care providers, and to Pediatrics Nationwide. He has served as a communications specialist at the Center for Injury Research and Policy at The Research Institute and came to Nationwide Children’s after 14-year career as daily newspaper reporter, most recently at The Columbus Dispatch.
-
Jeb Phillipshttps://pediatricsnationwide.org/author/jeb-phillips/October 13, 2015
-
Jeb Phillipshttps://pediatricsnationwide.org/author/jeb-phillips/
-
Jeb Phillipshttps://pediatricsnationwide.org/author/jeb-phillips/November 24, 2015
-
Jeb Phillipshttps://pediatricsnationwide.org/author/jeb-phillips/January 19, 2016
- Post Tags:
- Gastroenterology
- Neonatology
- Posted In:
- Features